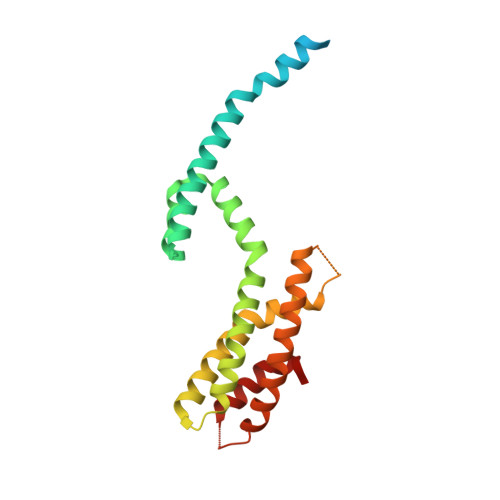
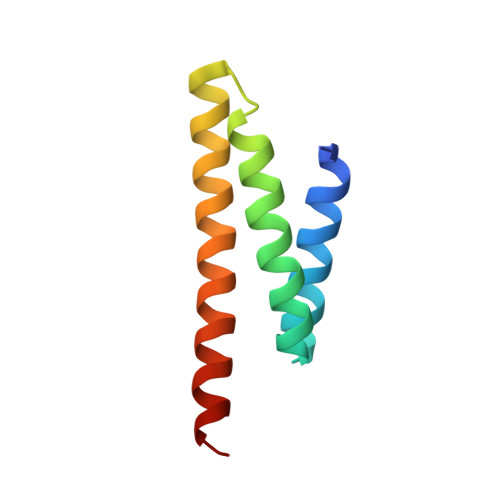
Crystal Structure of a Heterotetrameric Katanin p60:p80 Complex.
Faltova, L., Jiang, K., Frey, D., Wu, Y., Capitani, G., Prota, A.E., Akhmanova, A., Steinmetz, M.O., Kammerer, R.A.(2019) Structure 27: 1375-1383.e3
- PubMed: 31353241 Search on PubMed
- DOI: https://doi.org/10.1016/j.str.2019.07.002
- Primary Citation Related Structures:
6GZC - PubMed Abstract:
Katanin is a microtubule-severing enzyme that is crucial for many cellular processes. Katanin consists of two subunits, p60 and p80, that form a stable complex. The interaction between subunits is mediated by the p60 N-terminal microtubule-interacting and -trafficking domain (p60-MIT) and the p80 C-terminal domain (p80-CTD). Here, we performed a biophysical characterization of the mouse p60-MIT:p80-CTD heterodimer and show that this complex can assemble into heterotetramers. We identified two mutations that enhance heterotetramer formation and determined the X-ray crystal structure of this mutant complex. The structure revealed a domain-swapped heterotetramer consisting of two p60-MIT:p80-CTD heterodimers. Structure-based sequence alignments suggest that heterotetramerization of katanin might be a common feature of various species. Furthermore, we show that enhanced heterotetramerization of katanin impairs its microtubule end-binding properties and increases the enzyme's microtubule lattice binding and severing activities. Therefore, our findings suggest the existence of different katanin oligomers that possess distinct functional properties.
- Laboratory of Biomolecular Research, Division of Biology and Chemistry, Paul Scherrer Institut, 5232 Villigen PSI, Switzerland.
Organizational Affiliation: