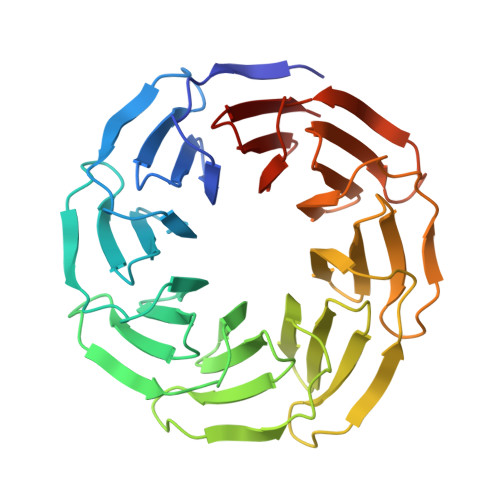
Computational design of symmetrical eight-bladed beta-propeller proteins.
Noguchi, H., Addy, C., Simoncini, D., Wouters, S., Mylemans, B., Van Meervelt, L., Schiex, T., Zhang, K.Y.J., Tame, J.R.H., Voet, A.R.D.(2019) IUCrJ 6: 46-55
- PubMed: 30713702 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1107/S205225251801480X
- Primary Citation Related Structures:
6G6M, 6G6N, 6G6O, 6G6P, 6G6Q - PubMed Abstract:
β-Propeller proteins form one of the largest families of protein structures, with a pseudo-symmetrical fold made up of subdomains called blades. They are not only abundant but are also involved in a wide variety of cellular processes, often by acting as a platform for the assembly of protein complexes. WD40 proteins are a subfamily of propeller proteins with no intrinsic enzymatic activity, but their stable, modular architecture and versatile surface have allowed evolution to adapt them to many vital roles. By computationally reverse-engineering the duplication, fusion and diversification events in the evolutionary history of a WD40 protein, a perfectly symmetrical homologue called Tako8 was made. If two or four blades of Tako8 are expressed as single polypeptides, they do not self-assemble to complete the eight-bladed architecture, which may be owing to the closely spaced negative charges inside the ring. A different computational approach was employed to redesign Tako8 to create Ika8, a fourfold-symmetrical protein in which neighbouring blades carry compensating charges. Ika2 and Ika4, carrying two or four blades per subunit, respectively, were found to assemble spontaneously into a complete eight-bladed ring in solution. These artificial eight-bladed rings may find applications in bionanotechnology and as models to study the folding and evolution of WD40 proteins.
- Laboratory of Biomolecular Modelling and Design, Department of Chemistry, KU Leuven, Celestijnenlaan 200G, 3001 Leuven, Belgium.
Organizational Affiliation: