A GenoChemetic strategy for derivatization of the violacein natural product scaffold
Lai, H.E., Obled, A.M.C., Chee, S.M., Morgan, R.M., Sharma, S.V., Moore, S.J., Polizzi, K.M., Goss, R.J.M., Freemont, P.S.(2019) bioRxiv
Experimental Data Snapshot
Starting Model: experimental
View more details
(2019) bioRxiv
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Flavin-dependent L-tryptophan oxidase VioA | A [auth B], B [auth A] | 417 | Chromobacterium violaceum ATCC 12472 | Mutation(s): 0 Gene Names: vioA, CV_3274 EC: 1.4.3.23 | 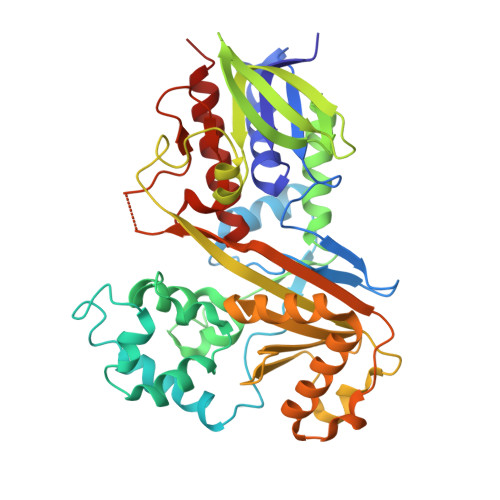 |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q9S3V1 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 3 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| FAD Download:Ideal Coordinates CCD File | C [auth B], F [auth A] | FLAVIN-ADENINE DINUCLEOTIDE C27 H33 N9 O15 P2 VWWQXMAJTJZDQX-UYBVJOGSSA-N |  | ||
| 4FW Download:Ideal Coordinates CCD File | D [auth B], G [auth A] | 4-FLUOROTRYPTOPHANE C11 H11 F N2 O2 DEBQMEYEKKWIKC-QMMMGPOBSA-N |  | ||
| MG Download:Ideal Coordinates CCD File | E [auth B], H [auth A] | MAGNESIUM ION Mg JLVVSXFLKOJNIY-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 151.812 | α = 90 |
| b = 174.88 | β = 90 |
| c = 93.919 | γ = 90 |
| Software Name | Purpose |
|---|---|
| PHENIX | refinement |
| xia2 | data reduction |
| Aimless | data scaling |
| PHASER | phasing |
| Funding Organization | Location | Grant Number |
|---|---|---|
| Imperial College London | United Kingdom | President's PhD Scholarship |