Structural conservation in a membrane-enveloped filamentous virus infecting a hyperthermophilic acidophile.
Liu, Y., Osinski, T., Wang, F., Krupovic, M., Schouten, S., Kasson, P., Prangishvili, D., Egelman, E.H.(2018) Nat Commun 9: 3360-3360
- PubMed: 30135568 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41467-018-05684-6
- Primary Citation Related Structures:
6D5F - PubMed Abstract:
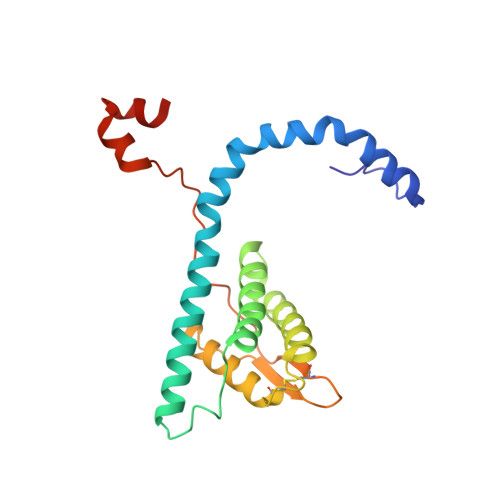
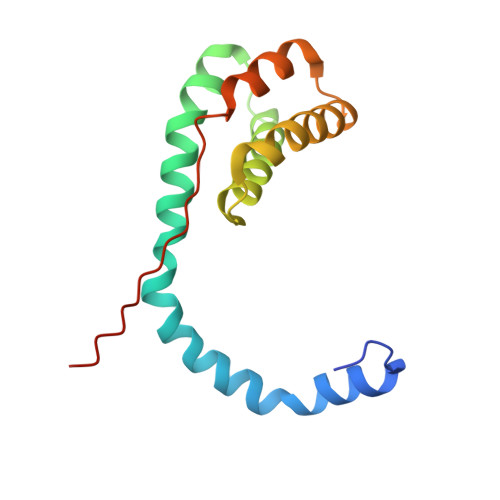
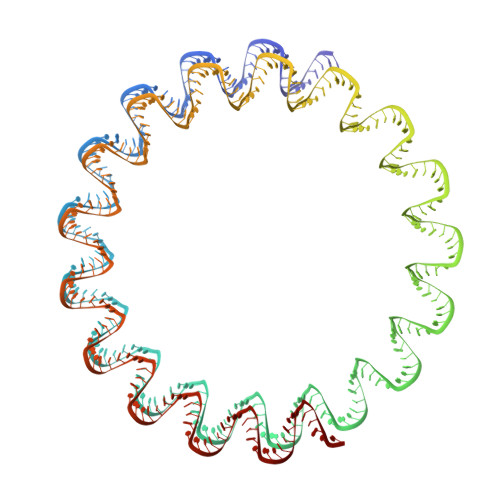
Different forms of viruses that infect archaea inhabiting extreme environments continue to be discovered at a surprising rate, suggesting that the current sampling of these viruses is sparse. We describe here Sulfolobus filamentous virus 1 (SFV1), a membrane-enveloped virus infecting Sulfolobus shibatae. The virus encodes two major coat proteins which display no apparent sequence similarity with each other or with any other proteins in databases. We have used cryo-electron microscopy at 3.7 Å resolution to show that these two proteins form a nearly symmetrical heterodimer, which wraps around A-form DNA, similar to what has been shown for SIRV2 and AFV1, two other archaeal filamentous viruses. The thin (∼ 20 Å) membrane of SFV1 is mainly archaeol, a lipid species that accounts for only 1% of the host lipids. Our results show how relatively conserved structural features can be maintained across evolution by both proteins and lipids that have diverged considerably.
- Institut Pasteur, Department of Microbiology, 25 rue du Dr. Roux, Paris, 75015, France.
Organizational Affiliation: