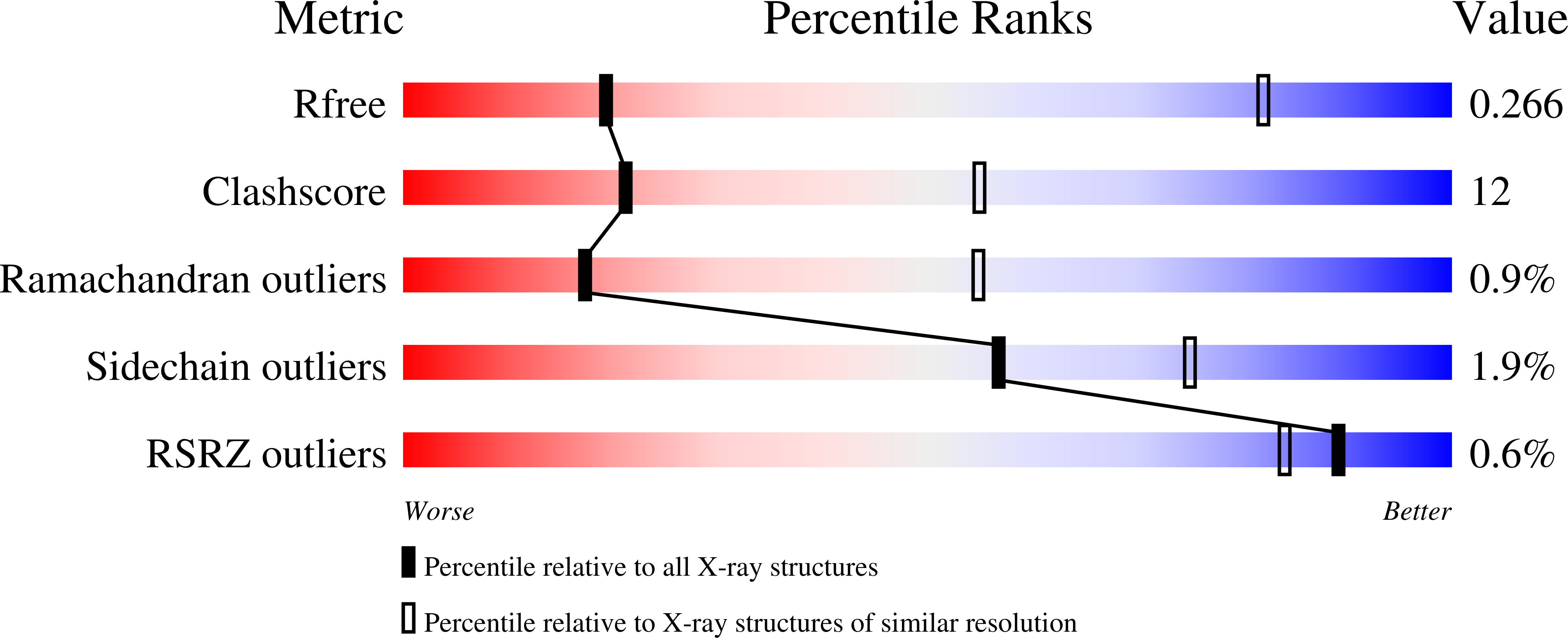
Kinetic mechanism of coupled binding in sodium-aspartate symporter GltPh.
Oh, S., Boudker, O.(2018) Elife 7
- PubMed: 30255846 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.7554/eLife.37291
- Primary Citation Related Structures:
6CTF - PubMed Abstract:
Many secondary active membrane transporters pump substrates against concentration gradients by coupling their uptake to symport of sodium ions. Symport requires the substrate and ions to be always transported together. Cooperative binding of the solutes is a key mechanism contributing to coupled transport in the sodium and aspartate symporter from Pyrococcus horikoshii Glt Ph . Here, we describe the kinetic mechanism of coupled binding for Glt Ph in the inward facing state. The first of the three coupled sodium ions, binds weakly and slowly, enabling the protein to accept the rest of the ions and the substrate. The last ion binds tightly, but is in rapid equilibrium with solution. Its release is required for the complex disassembly. Thus, the first ion serves to 'open the door' for the substrate, the last ion 'locks the door' once the substrate is in, and one ion contributes to both events.
- Department of Physiology & Biophysics, Weill Cornell Medicine, Cornell University, New York, United States.
Organizational Affiliation: