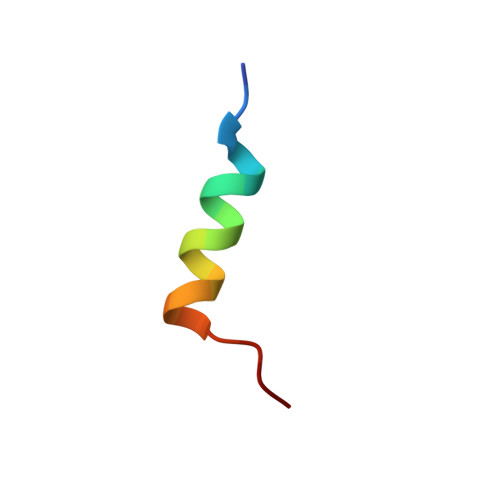
A Computationally Designed Peptide Derived from Escherichia coli as a Potential Drug Template for Antibacterial and Antibiofilm Therapies.
Cardoso, M.H., Candido, E.S., Chan, L.Y., Der Torossian Torres, M., Oshiro, K.G.N., Rezende, S.B., Porto, W.F., Lu, T.K., de la Fuente-Nunez, C., Craik, D.J., Franco, O.L.(2018) ACS Infect Dis 4: 1727-1736
- PubMed: 30346140 Search on PubMed
- DOI: https://doi.org/10.1021/acsinfecdis.8b00219
- Primary Citation Related Structures:
6CT4 - PubMed Abstract:
Computer-aided screening of antimicrobial peptides (AMPs) is a promising approach for discovering novel therapies against multidrug-resistant bacterial infections. Here, we functionally and structurally characterized an Escherichia coli-derived AMP (EcDBS1R5) previously designed through pattern identification [α-helical set (KK[ILV] (3) [AILV])], followed by sequence optimization. EcDBS1R5 inhibited the growth of Gram-negative and Gram-positive, susceptible and resistant bacterial strains at low doses (2-32 μM), with no cytotoxicity observed against non-cancerous and cancerous cell lines in the concentration range analyzed (<100 μM). Furthermore, EcDBS1R5 (16 μM) acted on Pseudomonas aeruginosa pre-formed biofilms by compromising the viability of biofilm-constituting cells. The in vivo antibacterial potential of EcDBS1R5 was confirmed as the peptide reduced bacterial counts by two-logs 2 days post-infection using a skin scarification mouse model. Structurally, circular dichroism analysis revealed that EcDBS1R5 is unstructured in hydrophilic environments, but has strong helicity in 2,2,2-trifluoroethanol (TFE)/water mixtures (v/v) and sodium dodecyl sulfate (SDS) micelles. The TFE-induced nuclear magnetic resonance structure of EcDBS1R5 was determined and showed an amphipathic helical segment with flexible termini. Moreover, we observed that the amide protons for residues Met2-Ala8, Arg10, Ala13-Ala16, and Trp19 in EcDBS1R5 are protected from the solvent, as their temperature coefficients values are more positive than -4.6 ppb·K -1 . In summary, this study reports a novel dual-antibacterial/antibiofilm α-helical peptide with therapeutic potential in vitro and in vivo against clinically relevant bacterial strains.
- Programa de Pós-Graduação em Patologia Molecular, Faculdade de Medicina , Universidade de Brasília , Campus Darcy Ribeiro, Asa Norte , Brasília , DF 70910900 , Brazil.
Organizational Affiliation: