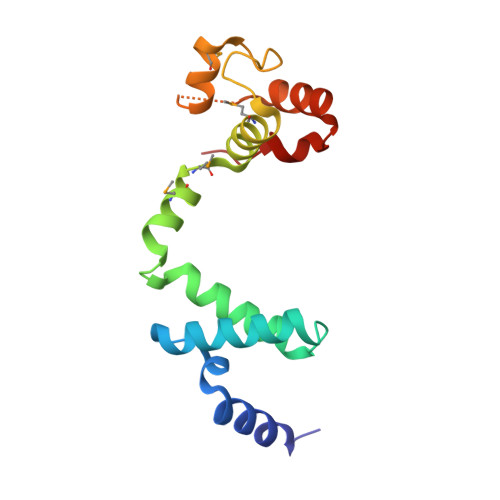
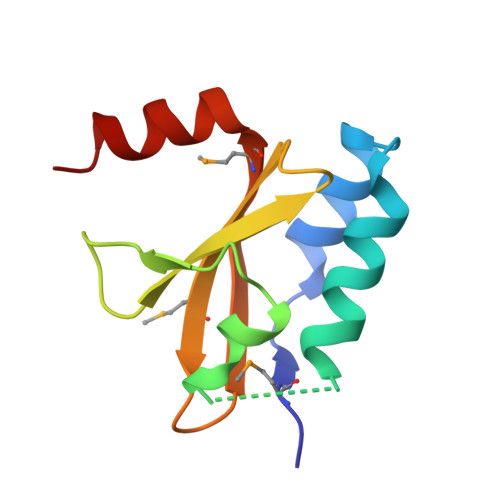
Structural changes of antitoxin HigA from Shigella flexneri by binding of its cognate toxin HigB.
Yoon, W.S., Seok, S.H., Won, H.S., Cho, T., Lee, S.J., Seo, M.D.(2019) Int J Biol Macromol 130: 99-108
- PubMed: 30797012 Search on PubMed
- DOI: https://doi.org/10.1016/j.ijbiomac.2019.02.111
- Primary Citation Related Structures:
5YCL, 6IRP - PubMed Abstract:
In toxin-antitoxin systems, many antitoxin proteins that neutralize their cognate toxin proteins also bind to DNA to repress transcription, and the DNA-binding affinity of the antitoxin is affected by its toxin. We solved crystal structures of the antitoxin HigA (apo- Sf HigA) and its complex with the toxin HigB ( Sf HigBA) from Shigella flexneri. The apo- Sf HigA shows a distinctive V-shaped homodimeric conformation with sequestered N-domains having a novel fold. Sf HigBA appears as a heterotetramer formed by N-terminal dimerization of Sf HigB-bound Sf HigA molecules. The conformational change in Sf HigA upon Sf HigB binding is mediated by rigid-body movements of its C-domains, which accompanied an overall conformational change from wide V-shaped to narrow V-shaped dimer. Consequently, the two putative DNA-binding helices (α7 in each subunit) are repositioned to a conformation more compatible with canonical homodimeric DNA-binding proteins containing HTH motifs. Collectively, this study demonstrates a conformational change in an antitoxin protein, which occurs upon toxin binding and is responsible for regulating antitoxin DNA binding.
- Department of Molecular Science and Technology, Ajou University, Suwon, Gyeonggi 16499, Republic of Korea; College of Pharmacy and Research Institute of Pharmaceutical Science and Technology (RIPST), Ajou University, Suwon, Gyeonggi 16499, Republic of Korea.
Organizational Affiliation: