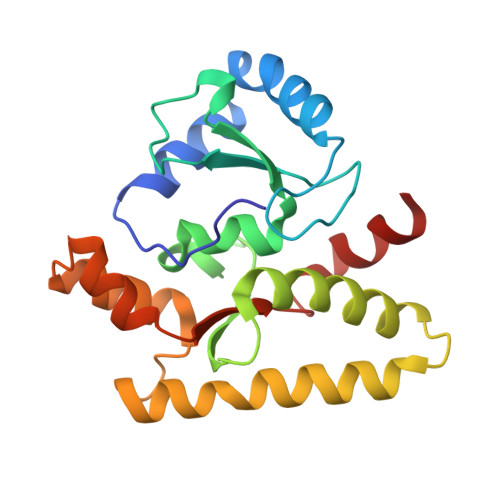
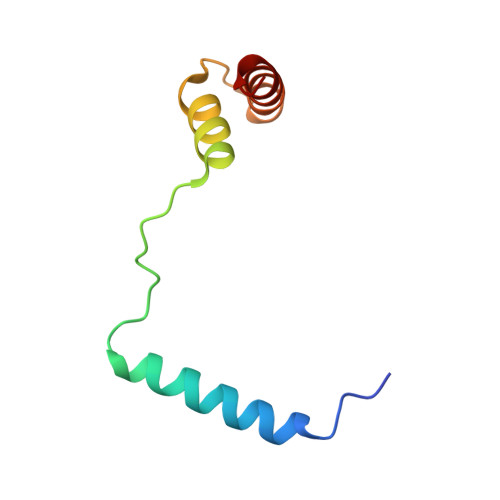
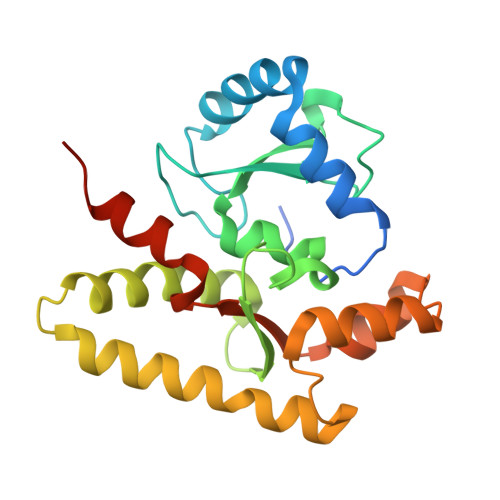
Architecture of the type IV coupling protein complex of Legionella pneumophila
Kwak, M.J., Kim, J.D., Kim, H., Kim, C., Bowman, J.W., Kim, S., Joo, K., Lee, J., Jin, K.S., Kim, Y.G., Lee, N.K., Jung, J.U., Oh, B.H.(2017) Nat Microbiol 2: 17114-17114
- PubMed: 28714967 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/nmicrobiol.2017.114
- Primary Citation Related Structures:
5X1E, 5X1H, 5X1U, 5X42, 5X90 - PubMed Abstract:
Many bacteria, including Legionella pneumophila, rely on the type IV secretion system to translocate a repertoire of effector proteins into the hosts for their survival and growth. Type IV coupling protein (T4CP) is a hexameric ATPase that links translocating substrates to the transenvelope secretion conduit. Yet, how a large number of effector proteins are selectively recruited and processed by T4CPs remains enigmatic. DotL, the T4CP of L. pneumophila, contains an ATPase domain and a C-terminal extension whose function is unknown. Unlike T4CPs involved in plasmid DNA translocation, DotL appeared to function by forming a multiprotein complex with four other proteins. Here, we show that the C-terminal extension of DotL interacts with DotN, IcmS, IcmW and an additionally identified subunit LvgA, and that this pentameric assembly binds Legionella effector proteins. We determined the crystal structure of this assembly and built an architecture of the T4CP holocomplex by combining a homology model of the ATPase domain of DotL. The holocomplex is a hexamer of a bipartite structure composed of a membrane-proximal ATPase domain and a membrane-distal substrate-recognition assembly. The presented information demonstrates the architecture and functional dissection of the multiprotein T4CP complexes and provides important insights into their substrate recruitment and processing.
- Department of Biological Sciences, KAIST Institute for the Biocentury, Korea Advanced Institute of Science and Technology, Daejeon 34141, Korea.
Organizational Affiliation: