Antibody Switch Residue Engineering for Improved Crystallization Chaperones
Bailey, L.J., Kossiakoff, A.A.To be published.
Experimental Data Snapshot
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Fab Heavy Chain | A [auth H] | 217 | Homo sapiens | Mutation(s): 0 | 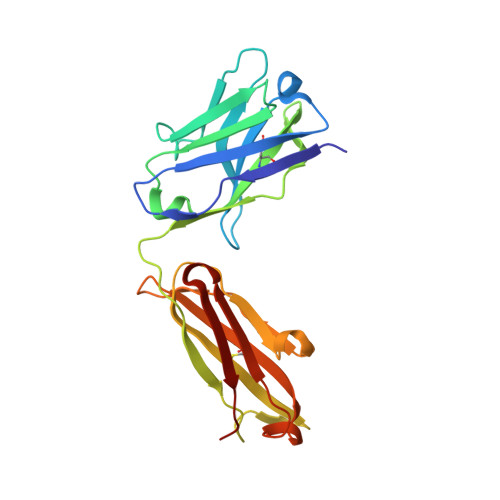 |
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Fab Light Chain | B [auth L] | 210 | Homo sapiens | Mutation(s): 0 | 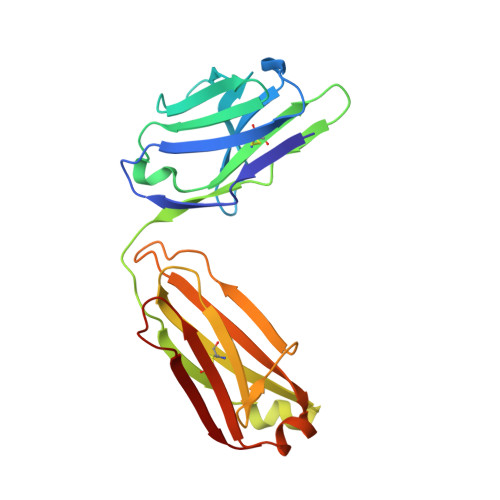 |
Entity ID: 3 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Histone chaperone ASF1 | C [auth B] | 153 | Saccharomyces cerevisiae S288C | Mutation(s): 0 Gene Names: ASF1, CIA1, YJL115W, J0755 | 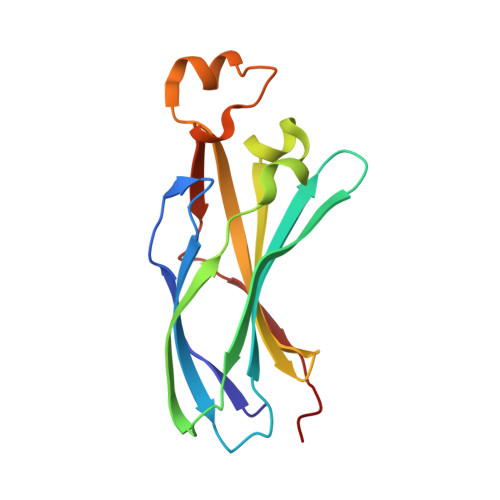 |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P32447 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 45.378 | α = 106.29 |
| b = 49.18 | β = 95.71 |
| c = 84.707 | γ = 100.13 |
| Software Name | Purpose |
|---|---|
| PHENIX | refinement |
| HKL-2000 | data reduction |
| SCALEPACK | data scaling |
| MOLREP | phasing |
| Funding Organization | Location | Grant Number |
|---|---|---|
| National Institutes of Health/National Institute of General Medical Sciences (NIH/NIGMS) | United States | GM 087519 |
| National Institutes of Health/National Institute of General Medical Sciences (NIH/NIGMS) | United States | GM 117372 |