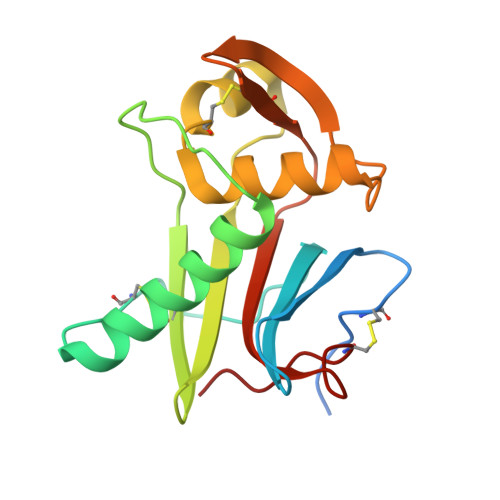
Mapping of the Lassa virus LAMP1 binding site reveals unique determinants not shared by other old world arenaviruses.
Israeli, H., Cohen-Dvashi, H., Shulman, A., Shimon, A., Diskin, R.(2017) PLoS Pathog 13: e1006337-e1006337
- PubMed: 28448640 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1371/journal.ppat.1006337
- Primary Citation Related Structures:
5NFF - PubMed Abstract:
Cell entry of many enveloped viruses occurs by engagement with cellular receptors, followed by internalization into endocytic compartments and pH-induced membrane fusion. A previously unnoticed step of receptor switching was found to be critical during cell entry of two devastating human pathogens: Ebola and Lassa viruses. Our recent studies revealed the functional role of receptor switching to LAMP1 for triggering membrane fusion by Lassa virus and showed the involvement of conserved histidines in this switching, suggesting that other viruses from this family may also switch to LAMP1. However, when we investigated viruses that are genetically close to Lassa virus, we discovered that they cannot bind LAMP1. A crystal structure of the receptor-binding module from Morogoro virus revealed structural differences that allowed mapping of the LAMP1 binding site to a unique set of Lassa residues not shared by other viruses in its family, illustrating a key difference in the cell-entry mechanism of Lassa virus that may contribute to its pathogenicity.
- Department of Structural Biology, Weizmann Institute of Science, Rehovot, Israel.
Organizational Affiliation: