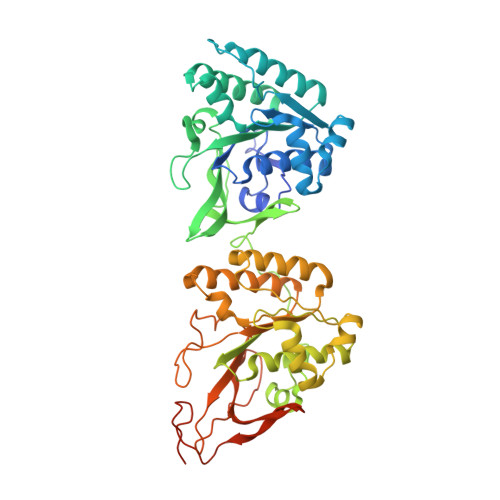
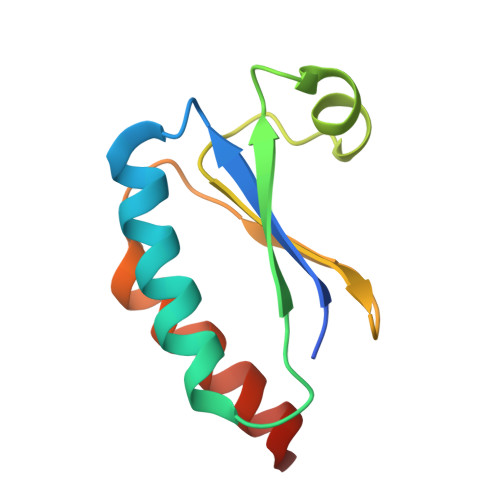
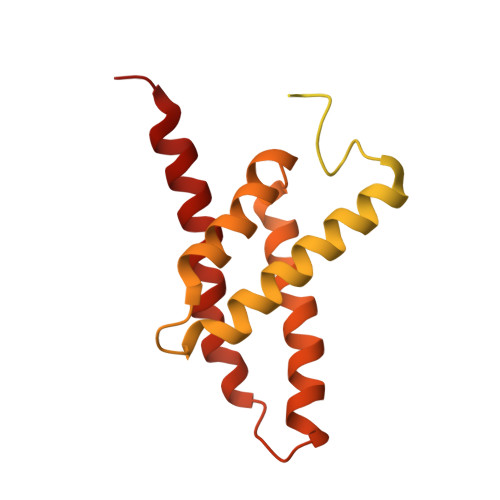
Structures of the cyanobacterial circadian oscillator frozen in a fully assembled state.
Snijder, J., Schuller, J.M., Wiegard, A., Lossl, P., Schmelling, N., Axmann, I.M., Plitzko, J.M., Forster, F., Heck, A.J.(2017) Science 355: 1181-1184
- PubMed: 28302852 Search on PubMed
- DOI: https://doi.org/10.1126/science.aag3218
- Primary Citation Related Structures:
5N8Y - PubMed Abstract:
Cyanobacteria have a robust circadian oscillator, known as the Kai system. Reconstituted from the purified protein components KaiC, KaiB, and KaiA, it can tick autonomously in the presence of adenosine 5'-triphosphate (ATP). The KaiC hexamers enter a natural 24-hour reaction cycle of autophosphorylation and assembly with KaiB and KaiA in numerous diverse forms. We describe the preparation of stoichiometrically well-defined assemblies of KaiCB and KaiCBA, as monitored by native mass spectrometry, allowing for a structural characterization by single-particle cryo-electron microscopy and mass spectrometry. Our data reveal details of the interactions between the Kai proteins and provide a structural basis to understand periodic assembly of the protein oscillator.
- Biomolecular Mass Spectrometry and Proteomics and Netherlands Proteomics Center, Bijvoet Center for Biomolecular Research, and Utrecht Institute for Pharmaceutical Sciences, Utrecht University, Padualaan 8, 3584 CH, Utrecht, Netherlands.
Organizational Affiliation: