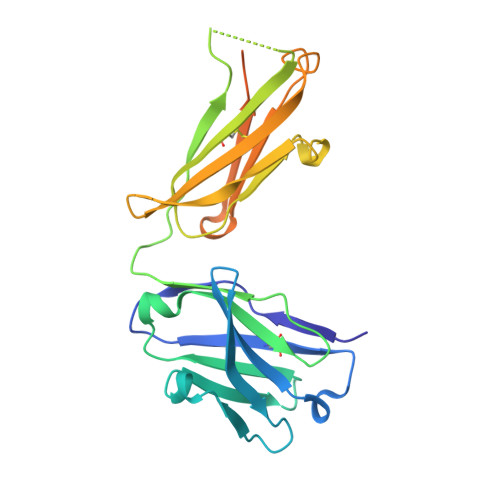
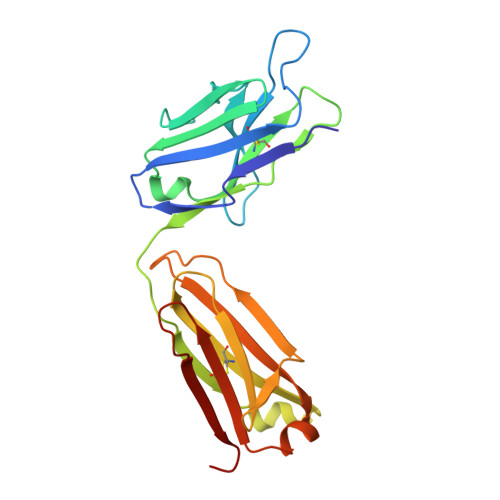
T cells control the generation of nanomolar-affinity anti-glycan antibodies.
Polonskaya, Z., Deng, S., Sarkar, A., Kain, L., Comellas-Aragones, M., McKay, C.S., Kaczanowska, K., Holt, M., McBride, R., Palomo, V., Self, K.M., Taylor, S., Irimia, A., Mehta, S.R., Dan, J.M., Brigger, M., Crotty, S., Schoenberger, S.P., Paulson, J.C., Wilson, I.A., Savage, P.B., Finn, M.G., Teyton, L.(2017) J Clin Invest 127: 1491-1504
- PubMed: 28287405 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1172/JCI91192
- Primary Citation Related Structures:
5JOP, 5JOR - PubMed Abstract:
Vaccines targeting glycan structures at the surface of pathogenic microbes must overcome the inherent T cell-independent nature of immune responses against glycans. Carbohydrate conjugate vaccines achieve this by coupling bacterial polysaccharides to a carrier protein that recruits heterologous CD4 T cells to help B cell maturation. Yet they most often produce low- to medium-affinity immune responses of limited duration in immunologically fit individuals and disappointing results in the elderly and immunocompromised patients. Here, we hypothesized that these limitations result from suboptimal T cell help. To produce the next generation of more efficacious conjugate vaccines, we have explored a synthetic design aimed at focusing both B cell and T cell recognition to a single short glycan displayed at the surface of a virus-like particle. We tested and established the proof of concept of this approach for 2 serotypes of Streptococcus pneumoniae. In both cases, these vaccines elicited serotype-specific, protective, and long-lasting IgG antibodies of nanomolar affinity against the target glycans in mice. We further identified a requirement for CD4 T cells in the anti-glycan antibody response. Our findings establish the design principles for improved glycan conjugate vaccines. We surmise that the same approach can be used for any microbial glycan of interest.