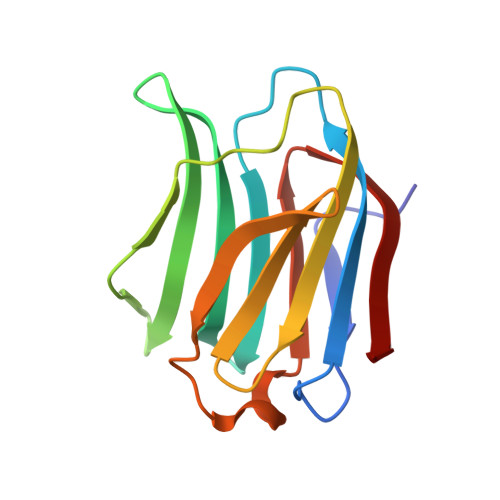
Controlling protein:ligand complex conformation through tuning of arginine-arene interactions: Synthetic and structural studies with 3-benzamido-2-sulfo-galactosides as galectin-3 ligands
Noresson, A.-L., Aurelius, O., Oberg, C.T., Engstrom, O., Sundin, A.P., Hakansson, M., Logan, D., Leffler, H., Nilsson, U.J.To be published.