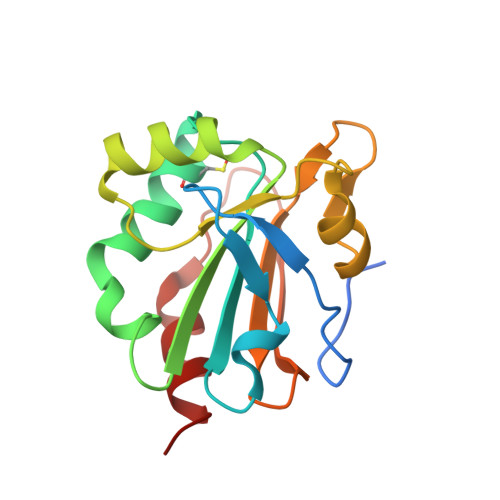
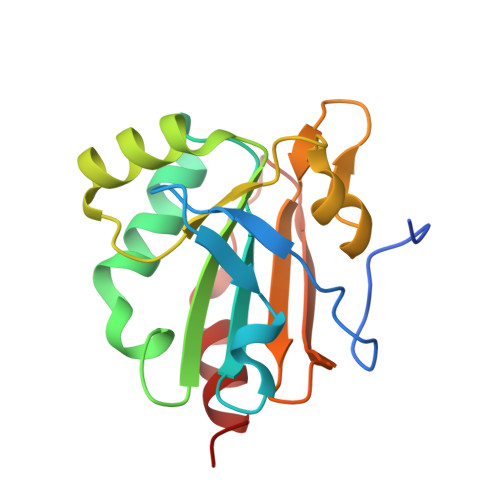
Redox chemistry of Mycobacterium tuberculosis alkylhydroperoxide reductase E (AhpE): Structural and mechanistic insight into a mycoredoxin-1 independent reductive pathway of AhpE via mycothiol
Kumar, A., Balakrishna, A.M., Nartey, W., Manimekalai, M.S.S., Gruber, G.(2016) Free Radic Biol Med 97: 588-601
- PubMed: 27417938 Search on PubMed
- DOI: https://doi.org/10.1016/j.freeradbiomed.2016.07.007
- Primary Citation Related Structures:
5ID2 - PubMed Abstract:
Mycobacterium tuberculosis (Mtb) has the ability to persist within the human host for a long time in a dormant stage and re-merges when the immune system is compromised. The pathogenic bacterium employs an elaborate antioxidant defence machinery composed of the mycothiol- and thioredoxin system in addition to a superoxide dismutase, a catalase, and peroxiredoxins (Prxs). Among the family of Peroxiredoxins, Mtb expresses a 1-cysteine peroxiredoxin, known as alkylhydroperoxide reductase E (MtAhpE), and defined as a potential tuberculosis drug target. The reduced MtAhpE (MtAhpE-SH) scavenges peroxides to become converted to MtAhpE-SOH. To provide continuous availability of MtAhpE-SH, MtAhpE-SOH has to become reduced. Here, we used NMR spectroscopy to delineate the reduced (MtAhpE-SH), sulphenic (MtAhpE-SOH) and sulphinic (MtAhpE-SO2H) states of MtAhpE through cysteinyl-labelling, and provide for the first time evidence of a mycothiol-dependent mechanism of MtAhpE reduction. This is confirmed by crystallographic studies, wherein MtAhpE was crystallized in the presence of mycothiol and the structure was solved at 2.43Å resolution. Combined with NMR-studies, the crystallographic structures reveal conformational changes of important residues during the catalytic cycle of MtAhpE. In addition, alterations of the overall protein in solution due to redox modulation are observed by small angle X-ray scattering (SAXS) studies. Finally, by employing SAXS and dynamic light scattering, insight is provided into the most probable physiological oligomeric state of MtAhpE necessary for activity, being also discussed in the context of concerted substrate binding inside the dimeric MtAhpE.
- Nanyang Technological University, School of Biological Sciences, 60 Nanyang Drive, Singapore 637551, Republic of Singapore.
Organizational Affiliation: