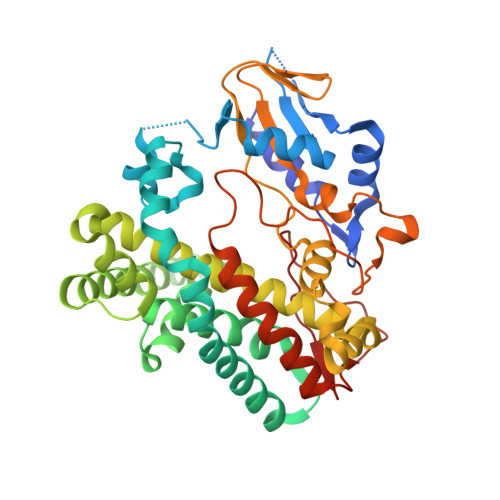
Structure of OxyAtei : completing our picture of the glycopeptide antibiotic producing Cytochrome P450 cascade.
Haslinger, K., Cryle, M.J.(2016) FEBS Lett 590: 571-581
- PubMed: 26820384 Search on PubMed
- DOI: https://doi.org/10.1002/1873-3468.12081
- Primary Citation Related Structures:
5HH3 - PubMed Abstract:
Cyclization of glycopeptide antibiotic precursors occurs in either three or four steps catalyzed by Cytochrome P450 enzymes. Three of these enzymes have been structurally characterized to date with the second enzyme along the pathway, OxyA, escaping structural analysis. We are now able to present the structure of OxyAtei involved in teicoplanin biosynthesis - the same enzyme recently shown to be the first active OxyA homolog. In spite of the hydrophobic character of the teicoplanin precursor, the polar active site of OxyAtei and its affinity for certain azole inhibitors hint at its preference for substrates with polar decorations.
- Max Planck Institute for Medical Research, Heidelberg, Germany.
Organizational Affiliation: