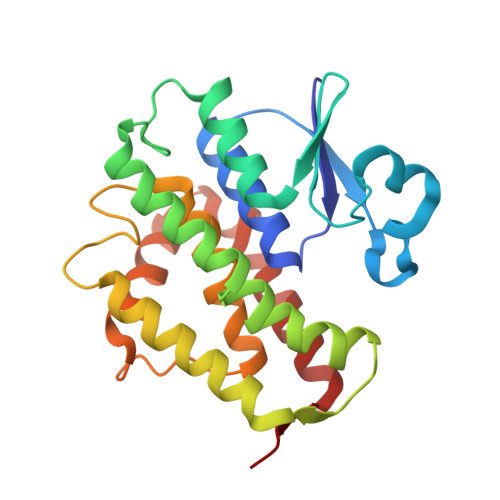
Epsilon glutathione transferases possess a unique class-conserved subunit interface motif that directly interacts with glutathione in the active site
Wongsantichon, J., Robinson, R.C., Ketterman, A.J.(2015) Biosci Rep 35
- PubMed: 26487708 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1042/BSR20150183
- Primary Citation Related Structures:
4YH2 - PubMed Abstract:
Epsilon class glutathione transferases (GSTs) have been shown to contribute significantly to insecticide resistance. We report a new Epsilon class protein crystal structure from Drosophila melanogaster for the glutathione transferase DmGSTE6. The structure reveals a novel Epsilon clasp motif that is conserved across hundreds of millions of years of evolution of the insect Diptera order. This histidine-serine motif lies in the subunit interface and appears to contribute to quaternary stability as well as directly connecting the two glutathiones in the active sites of this dimeric enzyme.
- Institute of Molecular and Cell Biology, A*STAR (Agency for Science, Technology and Research), Biopolis, Singapore 138673.
Organizational Affiliation: