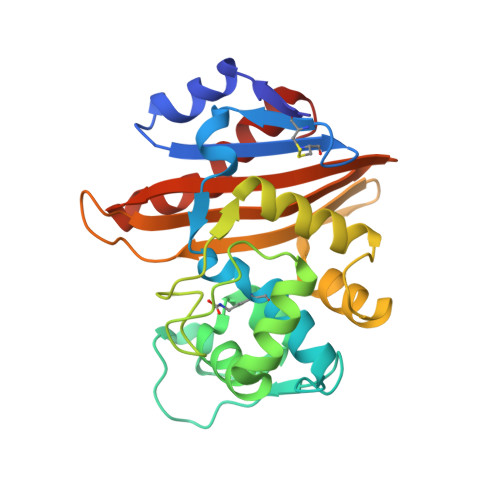
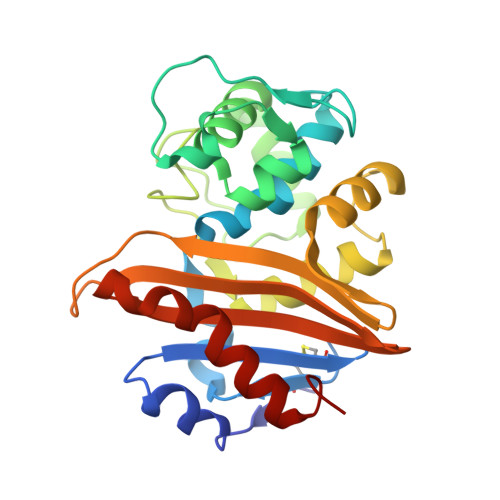
4,5-Disubstituted 6-Aryloxy-1,3-dihydrobenzo[c][1,2]oxaboroles Are Broad-Spectrum Serine beta-Lactamase Inhibitors.
McKinney, D.C., Zhou, F., Eyermann, C.J., Ferguson, A.D., Prince, D.B., Breen, J., Giacobbe, R.A., Lahiri, S., Verheijen, J.C.(2015) ACS Infect Dis 1: 310-316
- PubMed: 27622821 Search on PubMed
- DOI: https://doi.org/10.1021/acsinfecdis.5b00031
- Primary Citation Related Structures:
4WYY, 4WZ4, 4WZ5 - PubMed Abstract:
Bacterially expressed β-lactamases are rapidly eroding the clinical utility of the important β-lactam class of antibacterials, significantly impairing our ability to fight serious bacterial infections. This paper describes a study of oxaborole-derived β-lactamase inhibitors in which crystal structures and computational modeling aided in the rational design of analogues with improved spectrum of activity against class A, C, and D enzymes. Crystal structures of two of these inhibitors covalently bound to two different serine β-lactamases, class C Pseudomonas aeruginosa AmpC and class D OXA-10, are described herein. Improved physicochemical properties as well as increased activity against an array of β-lactamases resulted in substantial restoration of susceptibility to ceftazidime in Escherichia coli and Klebsiella pneumoniae.
- Infection Innovative Medicines, AstraZeneca R&D Boston , 35 Gatehouse Drive, Waltham Massachusetts 02451, United States.
Organizational Affiliation: