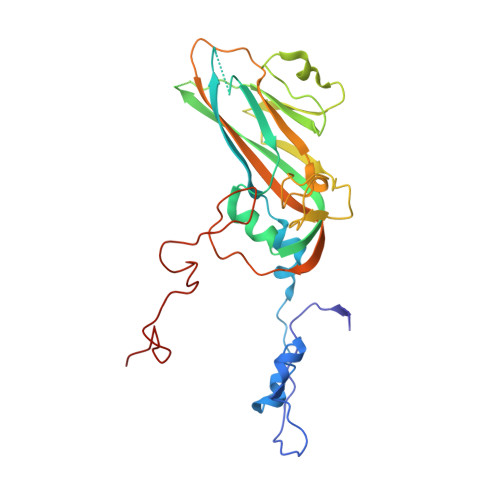
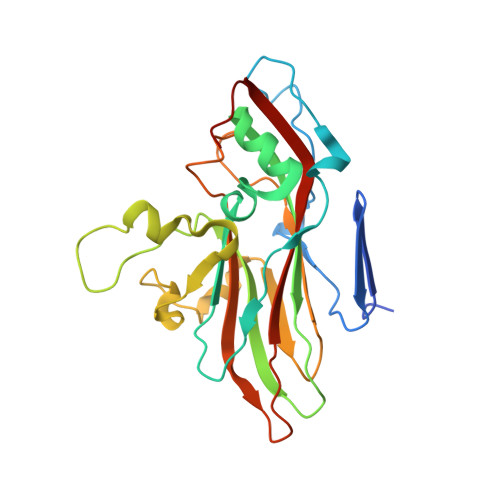
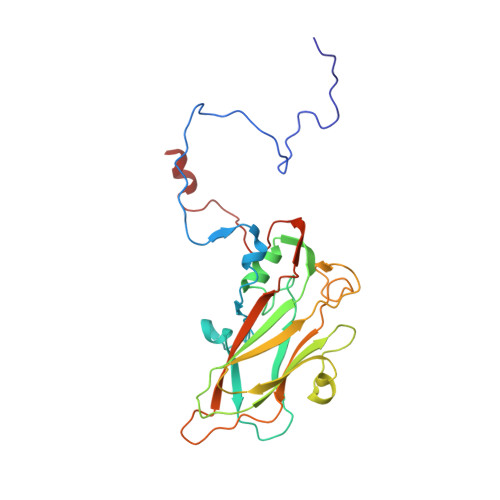
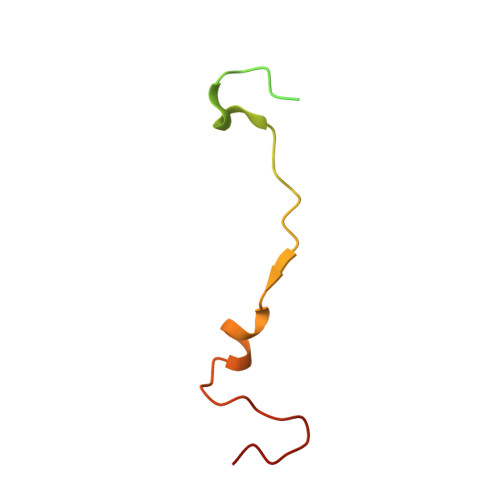
Virus structure. Structure and inhibition of EV-D68, a virus that causes respiratory illness in children.
Liu, Y., Sheng, J., Fokine, A., Meng, G., Shin, W.H., Long, F., Kuhn, R.J., Kihara, D., Rossmann, M.G.(2015) Science 347: 71-74
- PubMed: 25554786 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1126/science.1261962
- Primary Citation Related Structures:
4WM7, 4WM8 - PubMed Abstract:
Enterovirus D68 (EV-D68) is a member of Picornaviridae and is a causative agent of recent outbreaks of respiratory illness in children in the United States. We report here the crystal structures of EV-D68 and its complex with pleconaril, a capsid-binding compound that had been developed as an anti-rhinovirus drug. The hydrophobic drug-binding pocket in viral protein 1 contained density that is consistent with a fatty acid of about 10 carbon atoms. This density could be displaced by pleconaril. We also showed that pleconaril inhibits EV-D68 at a half-maximal effective concentration of 430 nanomolar and might, therefore, be a possible drug candidate to alleviate EV-D68 outbreaks.
- Department of Biological Sciences, Hockmeyer Hall of Structural Biology, 240 South Martin Jischke Drive, Purdue University, West Lafayette, IN 47907, USA.
Organizational Affiliation: