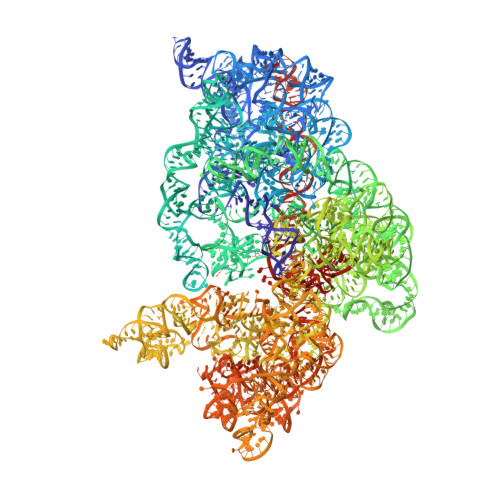
Crystal Structure of a 70S Ribosome-tRNA Complex Reveals Functional Interactions and Rearrangements
Korostelev, A., Trakhanov, S., Laurberg, M., Noller, H.F.(2006) Cell 126: 1065-1077
- PubMed: 16962654 Search on PubMed
- DOI: https://doi.org/10.1016/j.cell.2006.08.032
- Primary Citation Related Structures:
4V4I - PubMed Abstract:
Our understanding of the mechanism of protein synthesis has undergone rapid progress in recent years as a result of low-resolution X-ray and cryo-EM structures of ribosome functional complexes and high-resolution structures of ribosomal subunits and vacant ribosomes. Here, we present the crystal structure of the Thermus thermophilus 70S ribosome containing a model mRNA and two tRNAs at 3.7 A resolution. Many structural details of the interactions between the ribosome, tRNA, and mRNA in the P and E sites and the ways in which tRNA structure is distorted by its interactions with the ribosome are seen. Differences between the conformations of vacant and tRNA-bound 70S ribosomes suggest an induced fit of the ribosome structure in response to tRNA binding, including significant changes in the peptidyl-transferase catalytic site.
- Center for Molecular Biology of RNA and Department of Molecular, Cell and Developmental Biology, University of California, Santa Cruz, CA 95064, USA.
Organizational Affiliation: