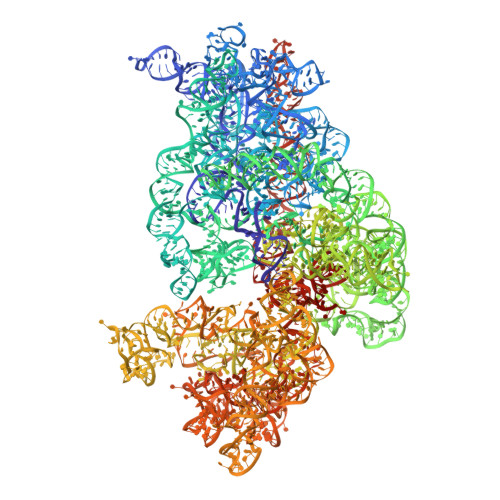
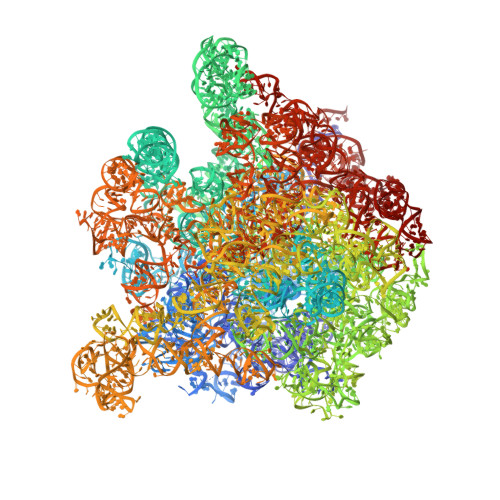
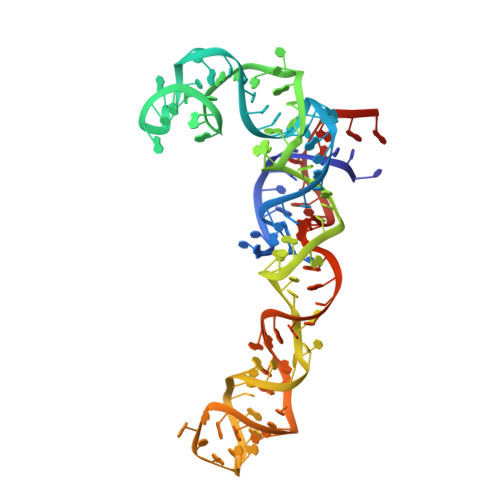
Structural basis for the control of translation initiation during stress.
Vila-Sanjurjo, A., Schuwirth, B.S., Hau, C.W., Cate, J.H.(2004) Nat Struct Mol Biol 11: 1054-1059
- PubMed: 15502846 Search on PubMed
- DOI: https://doi.org/10.1038/nsmb850
- Primary Citation Related Structures:
4V4G - PubMed Abstract:
During environmental stress, organisms limit protein synthesis by storing inactive ribosomes that are rapidly reactivated when conditions improve. Here we present structural and biochemical data showing that protein Y, an Escherichia coli stress protein, fills the tRNA- and mRNA-binding channel of the small ribosomal subunit to stabilize intact ribosomes. Protein Y inhibits translation initiation during cold shock but not at normal temperatures. Furthermore, protein Y competes with conserved translation initiation factors that, in bacteria, are required for ribosomal subunit dissociation. The mechanism used by protein Y to reduce translation initiation during stress and quickly release ribosomes for renewed translation initiation may therefore occur widely in nature.
- Department of Molecular and Cell Biology, University of California, Berkeley, California 94720, USA.
Organizational Affiliation: