Crystal structures of thermally stable adenylate kinase mutants designed by local structural entropy optimization and structure-guided mutagenesis
Moon, S., Bae, E.(2014) J Korean Soc Appl Biological Chem 57: 661-665
Experimental Data Snapshot
Starting Model: experimental
View more details
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Adenylate kinase | 217 | Bacillus subtilis subsp. subtilis str. 168 | Mutation(s): 38 Gene Names: adk, BSU01370 EC: 2.7.4.3 | 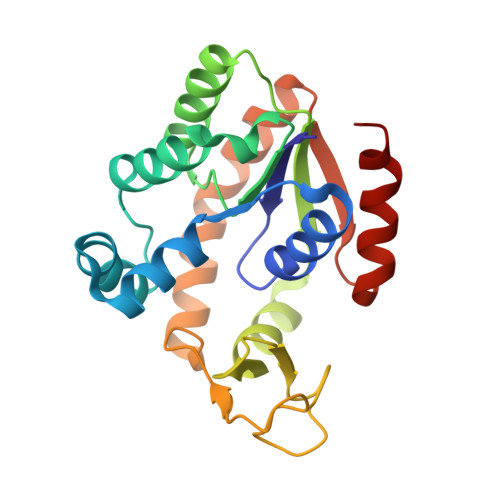 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P16304 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 4 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| AP5 Download:Ideal Coordinates CCD File | C [auth A], G [auth B] | BIS(ADENOSINE)-5'-PENTAPHOSPHATE C20 H29 N10 O22 P5 OIMACDRJUANHTJ-XPWFQUROSA-N |  | ||
| ZN Download:Ideal Coordinates CCD File | E [auth A], I [auth B] | ZINC ION Zn PTFCDOFLOPIGGS-UHFFFAOYSA-N |  | ||
| CA Download:Ideal Coordinates CCD File | F [auth A] | CALCIUM ION Ca BHPQYMZQTOCNFJ-UHFFFAOYSA-N |  | ||
| MG Download:Ideal Coordinates CCD File | D [auth A], H [auth B] | MAGNESIUM ION Mg JLVVSXFLKOJNIY-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 39.003 | α = 90.98 |
| b = 48.79 | β = 97.34 |
| c = 54.832 | γ = 95.83 |
| Software Name | Purpose |
|---|---|
| REFMAC | refinement |