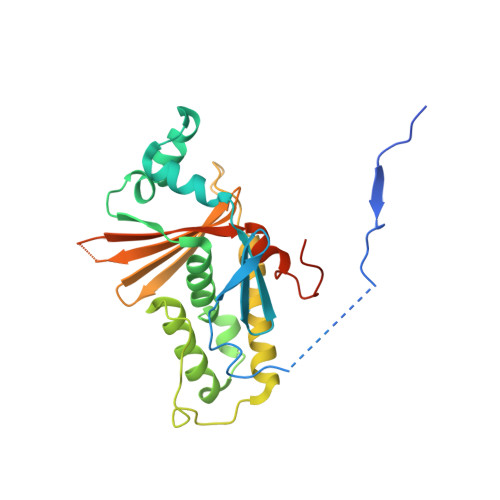
Crystal Structure of Marburg Virus VP24.
Zhang, A.P., Bornholdt, Z.A., Abelson, D.M., Saphire, E.O.(2014) J Virol 88: 5859-5863
- PubMed: 24574400 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1128/JVI.03565-13
- Primary Citation Related Structures:
4OR8 - PubMed Abstract:
The VP24 protein plays an essential, albeit poorly understood role in the filovirus life cycle. VP24 is only 30% identical between Marburg virus and the ebolaviruses. Furthermore, VP24 from the ebolaviruses is immunosuppressive, while that of Marburg virus is not. The crystal structure of Marburg virus VP24, presented here, reveals that although the core is similar between the viral genera, Marburg VP24 is distinguished by a projecting β-shelf and an alternate conformation of the N-terminal polypeptide.
- Department of Immunology and Microbial Science, The Scripps Research Institute, La Jolla, California, USA.
Organizational Affiliation: