Crystal structure of orotidine 5'-monophosphate decarboxylase from Methanobacterium thermoautotrophicum complexed with 3-deazauridine 5'-monophosphate
Fedorov, A.A., Fedorov, E.V., Iiams, V., Gerlt, J.A., Almo, S.C.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Orotidine 5'-phosphate decarboxylase | 228 | Methanothermobacter thermautotrophicus | Mutation(s): 1 Gene Names: pyrF, MTH_129 EC: 4.1.1.23 | 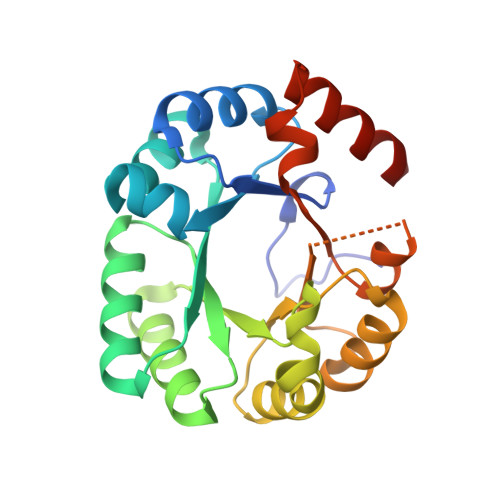 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | O26232 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 3 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| 3DU Download:Ideal Coordinates CCD File | C [auth A], F [auth B] | 4-hydroxy-1-(5-O-phosphono-beta-D-ribofuranosyl)pyridin-2(1H)-one C10 H14 N O9 P BMAAJONJGSMHCR-PEBGCTIMSA-N |  | ||
| PEG Download:Ideal Coordinates CCD File | E [auth A] | DI(HYDROXYETHYL)ETHER C4 H10 O3 MTHSVFCYNBDYFN-UHFFFAOYSA-N |  | ||
| CA Download:Ideal Coordinates CCD File | D [auth A], G [auth B] | CALCIUM ION Ca BHPQYMZQTOCNFJ-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 56.886 | α = 90 |
| b = 56.896 | β = 90 |
| c = 125.179 | γ = 90 |
| Software Name | Purpose |
|---|---|
| CBASS | data collection |
| BALBES | phasing |
| PHENIX | refinement |
| HKL-2000 | data reduction |
| HKL-2000 | data scaling |