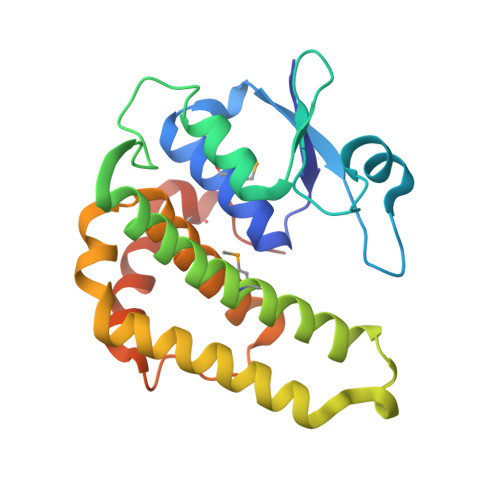
Crystal structure of a glutathione transferase family member from Psuedomonas fluorescens Pf-5, target EFI-900011, with bound S-(propanoic acid)-glutathione
Vetting, M.W., Sauder, J.M., Morisco, L.L., Wasserman, S.R., Sojitra, S., Imker, H.J., Burley, S.K., Gerlt, J.A., Almo, S.C., Enzyme Function Initiative (EFI)To be published.