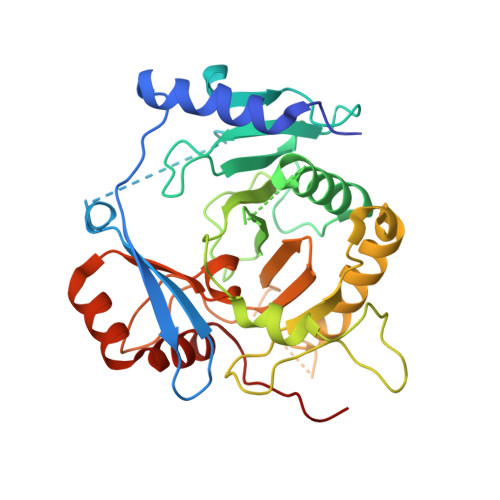
Structures of yeast Apa2 reveal catalytic insights into a canonical AP4A phosphorylase of the histidine triad superfamily
Hou, W.T., Li, W.Z., Chen, Y., Jiang, Y.L., Zhou, C.Z.(2013) J Mol Biology 425: 2687-2698
- PubMed: 23628156 Search on PubMed
- DOI: https://doi.org/10.1016/j.jmb.2013.04.018
- Primary Citation Related Structures:
4I5T, 4I5V, 4I5W - PubMed Abstract:
The homeostasis of intracellular diadenosine 5',5″'-P(1),P(4)-tetraphosphate (Ap4A) in the yeast Saccharomyces cerevisiae is maintained by two 60% sequence-identical paralogs of Ap4A phosphorylases (Apa1 and Apa2). Enzymatic assays show that, compared to Apa1, Apa2 has a relatively higher phosphorylase activity towards Ap3A (5',5″'-P(1),P(3)-tetraphosphate), Ap4A, and Ap5A (5',5″'-P(1),P(5)-tetraphosphate), and Ap4A is the favorable substrate for both enzymes. To decipher the catalytic insights, we determined the crystal structures of Apa2 in the apo-, AMP-, and Ap4A-complexed forms at 2.30, 2.80, and 2.70Å resolution, respectively. Apa2 is an α/β protein with a core domain of a twisted eight-stranded antiparallel β-sheet flanked by several α-helices, similar to the galactose-1-phosphate uridylyltransferase (GalT) members of the histidine triad (HIT) superfamily. However, a unique auxiliary domain enables an individual Apa2 monomer to possess an intact substrate-binding cleft, which is distinct from previously reported dimeric GalT proteins. This cleft is perfectly complementary to the favorable substrate Ap4A, the AMP and ATP moieties of which are perpendicular to each other, leaving the α-phosphate group exposed at the sharp turn against the catalytic residue His161. Structural comparisons combined with site-directed mutagenesis and activity assays enable us to define the key residues for catalysis. Furthermore, multiple-sequence alignment reveals that Apa2 and homologs represent canonical Ap4A phosphorylases, which could be grouped as a unique branch in the GalT family.
- Hefei National Laboratory for Physical Sciences at the Microscale and School of Life Sciences, University of Science and Technology of China, Hefei Anhui 230027, People's Republic of China.
Organizational Affiliation: