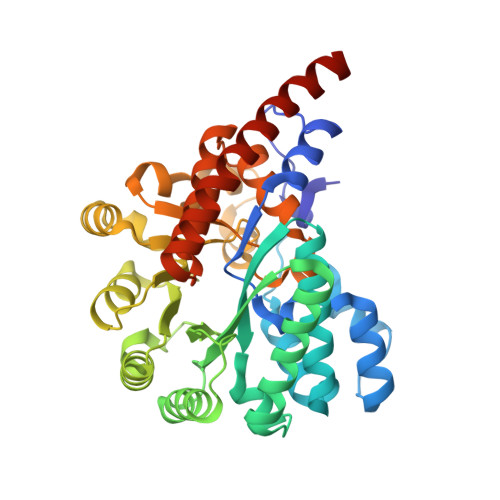
Crystal structure of a cog1816 amidohydrolase (target EFI-505188) from Burkhoderia ambifaria, with bound Zn
Vetting, M.W., Goble, A.M., Morisco, L.L., Wasserman, S.R., Sojitra, S., Imker, H.J., Raushel, F.M., Gerlt, J.A., Almo, S.C., Enzyme Function Initiative (EFI)To be published.