Crystal Structure of Putative Thioredoxin Reductase TrxB from Bacillus anthracis
Maltseva, N., Kim, Y., Kwon, K., Anderson, W.F., Joachimiak, A., Center for Structural Genomics of Infectious Diseases (CSGID)To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Putative thioredoxin reductase | 304 | Bacillus anthracis str. 'Ames Ancestor | Mutation(s): 0 Gene Names: BA_2768, GBAA_2768 | 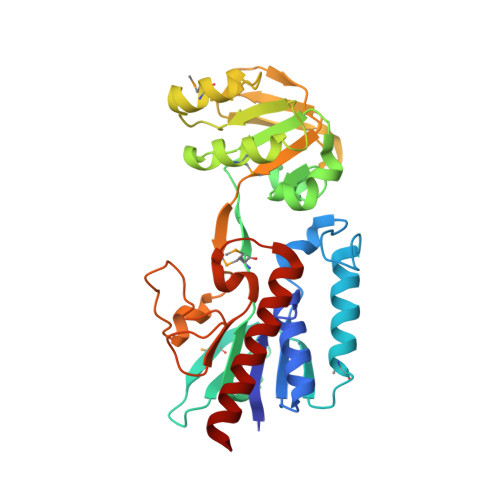 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A0A6L8PLA6 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Putative thioredoxin reductase | 304 | Bacillus anthracis str. 'Ames Ancestor | Mutation(s): 0 Gene Names: BA_2768, GBAA_2768 | 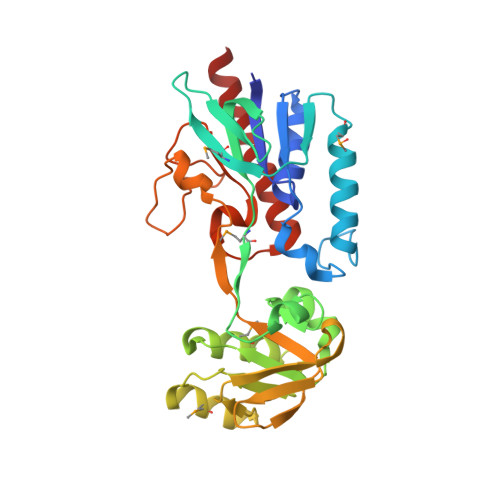 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A0A6L8PLA6 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 3 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| FAD Download:Ideal Coordinates CCD File | E [auth A], G [auth B], H [auth C], K [auth D] | FLAVIN-ADENINE DINUCLEOTIDE C27 H33 N9 O15 P2 VWWQXMAJTJZDQX-UYBVJOGSSA-N |  | ||
| GOL Download:Ideal Coordinates CCD File | M [auth D] | GLYCEROL C3 H8 O3 PEDCQBHIVMGVHV-UHFFFAOYSA-N |  | ||
| MG Download:Ideal Coordinates CCD File | F [auth A], I [auth C], J [auth C], L [auth D] | MAGNESIUM ION Mg JLVVSXFLKOJNIY-UHFFFAOYSA-N |  | ||
| Modified Residues 2 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Type | Formula | 2D Diagram | Parent |
| MSE Query on MSE | A, B, D | L-PEPTIDE LINKING | C5 H11 N O2 Se |  | MET |
| MSO Query on MSO | C | L-PEPTIDE LINKING | C5 H11 N O3 Se |  | MET |
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 82.558 | α = 90 |
| b = 54.096 | β = 93.04 |
| c = 140.037 | γ = 90 |
| Software Name | Purpose |
|---|---|
| SBC-Collect | data collection |
| HKL-3000 | data collection |
| HKL-3000 | phasing |
| MOLREP | phasing |
| PHENIX | refinement |
| HKL-3000 | data reduction |
| HKL-3000 | data scaling |