A Detailed Look Into Chinese Hamster Legumain Active Site Structure and Exploration of its Function
Li, W., Damme, M., Buessow, K., Grimm, I., Van Den Heuvel, J., Heinz, D.W., Krausze, J.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| PROLEGUMAIN | 408 | Cricetulus griseus | Mutation(s): 0 EC: 3.4.22.34 | 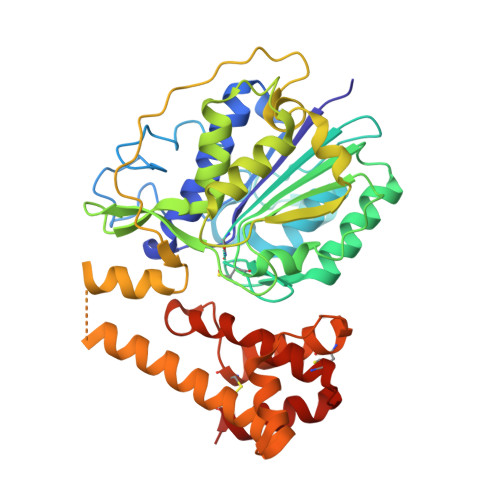 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | G3I1H5 | ||||
Glycosylation | |||||
| Glycosylation Sites: 3 | |||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| NAG Download:Ideal Coordinates CCD File | B [auth A], C [auth A], D [auth A] | 2-acetamido-2-deoxy-beta-D-glucopyranose C8 H15 N O6 OVRNDRQMDRJTHS-FMDGEEDCSA-N |  | ||
| Modified Residues 2 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Type | Formula | 2D Diagram | Parent |
| CSX Query on CSX | A | L-PEPTIDE LINKING | C3 H7 N O3 S |  | CYS |
| SNN Query on SNN | A | L-PEPTIDE LINKING | C4 H6 N2 O2 |  | ASN |
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 50.81 | α = 90 |
| b = 50.81 | β = 90 |
| c = 253.59 | γ = 120 |
| Software Name | Purpose |
|---|---|
| PHENIX | refinement |
| XDS | data reduction |
| XDS | data scaling |
| PHASER | phasing |