Structure of human XLF-XRCC4: assembly of a functional DNA repair complex
Andres, N.S., Wyman, C., Modesti, M., Junop, M.S.To be published.
Experimental Data Snapshot
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| DNA repair protein XRCC4 | 163 | Homo sapiens | Mutation(s): 0 Gene Names: XRCC4 | 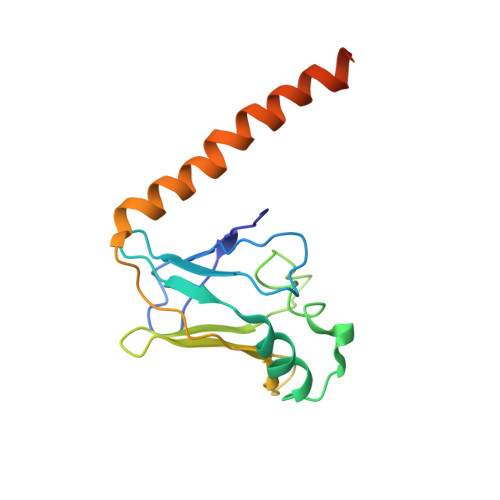 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: Q13426 GTEx: ENSG00000152422 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q13426 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Non-homologous end-joining factor 1 | 230 | Homo sapiens | Mutation(s): 0 Gene Names: NHEJ1, XLF | 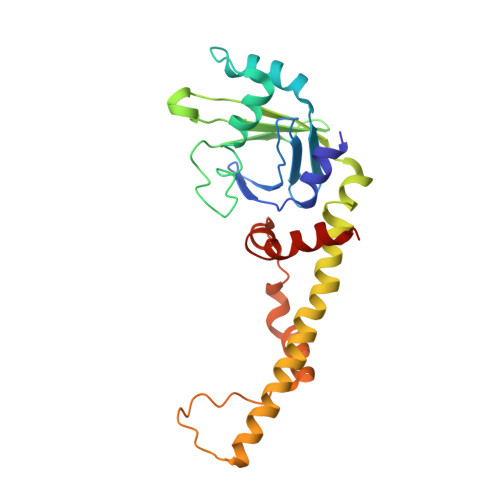 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: Q9H9Q4 GTEx: ENSG00000187736 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q9H9Q4 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| TBR Download:Ideal Coordinates CCD File | AA [auth J] BA [auth M] CA [auth P] DA [auth V] EA [auth X] | HEXATANTALUM DODECABROMIDE Br12 Ta6 YWYIQTPPCOBSGN-UHFFFAOYSA-M |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 745.379 | α = 90 |
| b = 149.585 | β = 94.72 |
| c = 80.475 | γ = 90 |
| Software Name | Purpose |
|---|---|
| PHENIX | refinement |
| PHENIX | model building |
| CNS | refinement |
| HKL-2000 | data reduction |
| HKL-2000 | data scaling |
| PHENIX | phasing |
| CBASS | data collection |