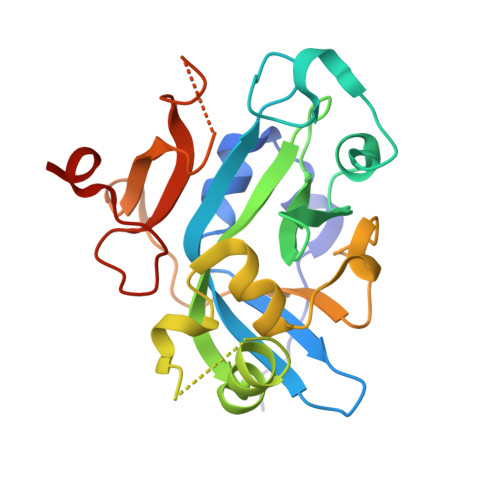
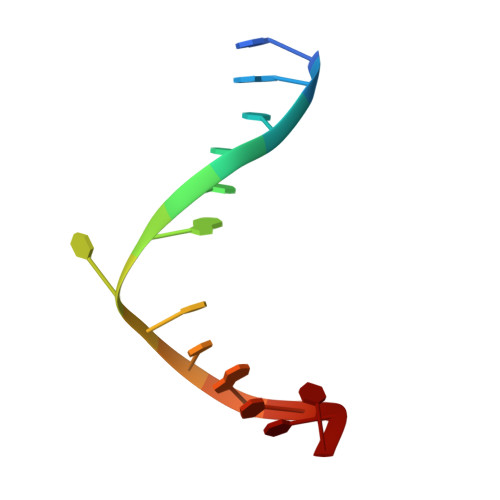
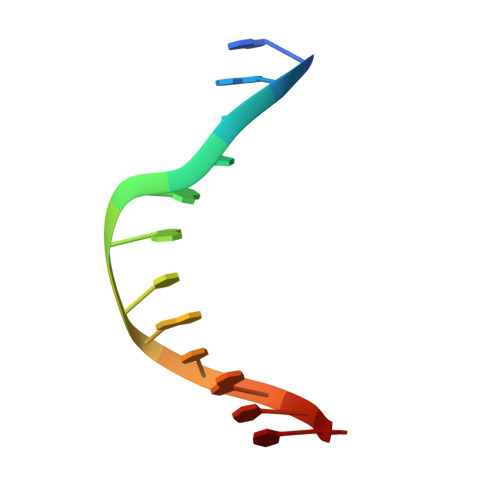
Structural Basis for the Inhibition of Human Alkyladenine DNA Glycosylase (AAG) by 3,N4-Ethenocytosine-containing DNA.
Lingaraju, G.M., Davis, C.A., Setser, J.W., Samson, L.D., Drennan, C.L.(2011) J Biological Chem 286: 13205-13213
- PubMed: 21349833 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1074/jbc.M110.192435
- Primary Citation Related Structures:
3QI5 - PubMed Abstract:
Reactive oxygen and nitrogen species, generated by neutrophils and macrophages in chronically inflamed tissues, readily damage DNA, producing a variety of potentially genotoxic etheno base lesions; such inflammation-related DNA damage is now known to contribute to carcinogenesis. Although the human alkyladenine DNA glycosylase (AAG) can specifically bind DNA containing either 1,N(6)-ethenoadenine (εA) lesions or 3,N(4)-ethenocytosine (εC) lesions, it can only excise εA lesions. AAG binds very tightly to DNA containing εC lesions, forming an abortive protein-DNA complex; such binding not only shields εC from repair by other enzymes but also inhibits AAG from acting on other DNA lesions. To understand the structural basis for inhibition, we have characterized the binding of AAG to DNA containing εC lesions and have solved a crystal structure of AAG bound to a DNA duplex containing the εC lesion. This study provides the first structure of a DNA glycosylase in complex with an inhibitory base lesion that is induced endogenously and that is also induced upon exposure to environmental agents such as vinyl chloride. We identify the primary cause of inhibition as a failure to activate the nucleotide base as an efficient leaving group and demonstrate that the higher binding affinity of AAG for εC versus εA is achieved through formation of an additional hydrogen bond between Asn-169 in the active site pocket and the O(2) of εC. This structure provides the basis for the design of AAG inhibitors currently being sought as an adjuvant for cancer chemotherapy.
- Center for Environmental Health Sciences, Massachusetts Institute of Technology, Cambridge, Massachusetts 02139, USA.
Organizational Affiliation: