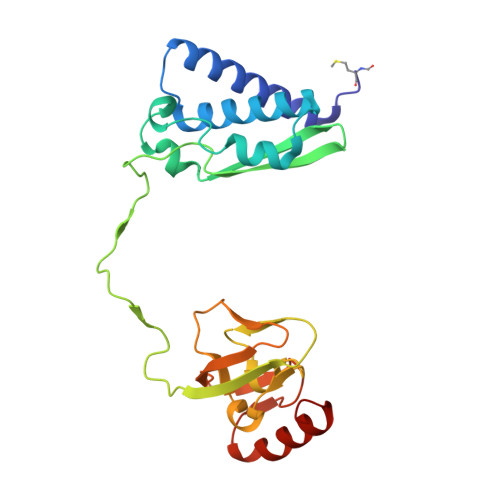
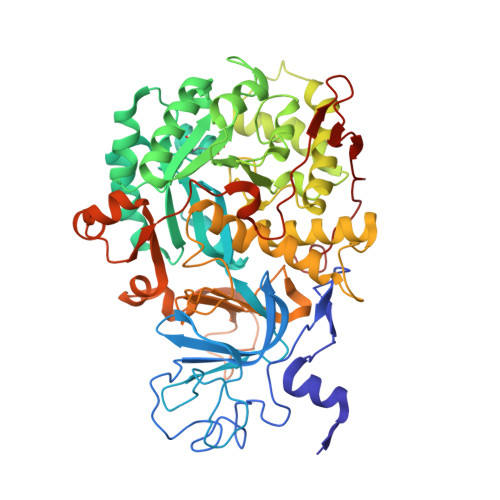
Iron-containing urease in a pathogenic bacterium.
Carter, E.L., Tronrud, D.E., Taber, S.R., Karplus, P.A., Hausinger, R.P.(2011) Proc Natl Acad Sci U S A 108: 13095-13099
- PubMed: 21788478 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1073/pnas.1106915108
- Primary Citation Related Structures:
3QGA, 3QGK - PubMed Abstract:
Helicobacter mustelae, a gastric pathogen of ferrets, synthesizes a distinct iron-dependent urease in addition to its archetypical nickel-containing enzyme. The iron-urease is oxygen-labile, with the inactive protein exhibiting a methemerythrin-like electronic spectrum. Significantly, incubation of the oxidized protein with dithionite under anaerobic conditions leads to restoration of activity and bleaching of the spectrum. Structural analysis of the oxidized species reveals a dinuclear iron metallocenter bridged by a lysine carbamate, closely resembling the traditional nickel-urease active site. Although the iron-urease is less active than the nickel-enzyme, its activity allows H. mustelae to survive the carnivore's low-nickel gastric environment.
- Department of Microbiology and Molecular Genetics, Michigan State University, East Lansing, MI 48824, USA.
Organizational Affiliation: