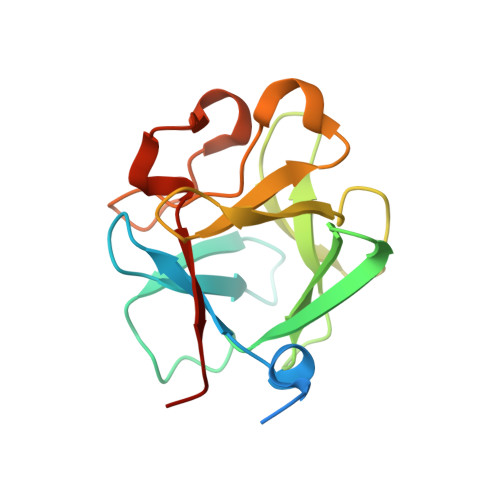
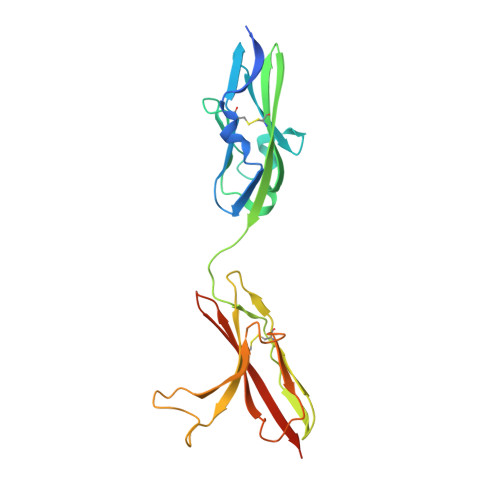
Plasticity in Interactions of Fibroblast Growth Factor 1 (FGF1) N Terminus with FGF Receptors Underlies Promiscuity of FGF1.
Beenken, A., Eliseenkova, A.V., Ibrahimi, O.A., Olsen, S.K., Mohammadi, M.(2012) J Biological Chem 287: 3067-3078
- PubMed: 22057274 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1074/jbc.M111.275891
- Primary Citation Related Structures:
3OJ2, 3OJM, 3OJV - PubMed Abstract:
Tissue-specific alternative splicing in the second half of Ig-like domain 3 (D3) of fibroblast growth factor receptors 1-3 (FGFR1 to -3) generates epithelial FGFR1b-FGFR3b and mesenchymal FGFR1c-FGFR3c splice isoforms. This splicing event establishes a selectivity filter to restrict the ligand binding specificity of FGFRb and FGFRc isoforms to mesenchymally and epithelially derived fibroblast growth factors (FGFs), respectively. FGF1 is termed the "universal FGFR ligand" because it overrides this specificity barrier. To elucidate the molecular basis for FGF1 cross-reactivity with the "b" and "c" splice isoforms of FGFRs, we determined the first crystal structure of FGF1 in complex with an FGFRb isoform, FGFR2b, at 2.1 Å resolution. Comparison of the FGF1-FGFR2b structure with the three previously published FGF1-FGFRc structures reveals that plasticity in the interactions of the N-terminal region of FGF1 with FGFR D3 is the main determinant of FGF1 cross-reactivity with both isoforms of FGFRs. In support of our structural data, we demonstrate that substitution of three N-terminal residues (Gly-19, His-25, and Phe-26) of FGF2 (a ligand that does not bind FGFR2b) for the corresponding residues of FGF1 (Phe-16, Asn-22, and Tyr-23) enables the FGF2 triple mutant to bind and activate FGFR2b. These findings taken together with our previous structural data on receptor binding specificity of FGF2, FGF8, and FGF10 conclusively show that sequence divergence at the N termini of FGFs is the primary regulator of the receptor binding specificity and promiscuity of FGFs.
- Department of Pharmacology, New York University School of Medicine, New York, New York 10016, USA.
Organizational Affiliation: