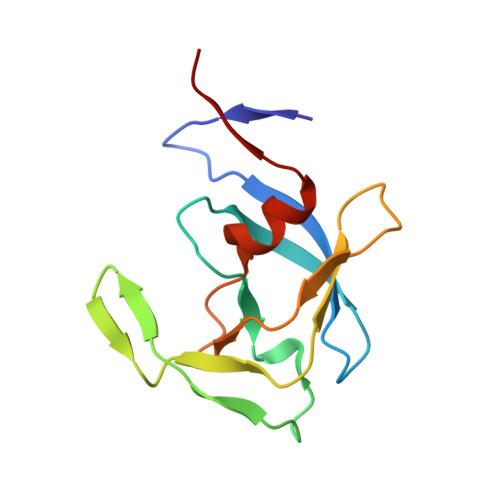
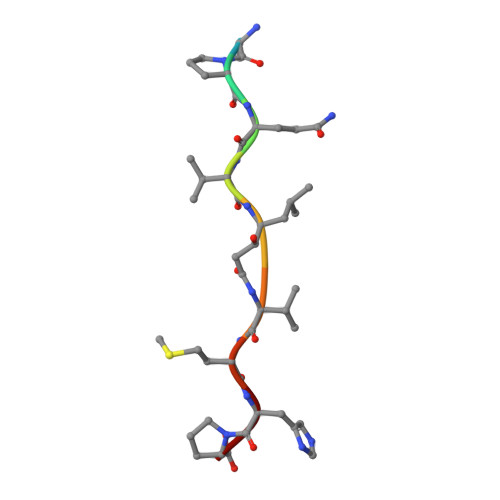
Crystal structures of inhibitor complexes of human T-cell leukemia virus (HTLV-1) protease.
Satoh, T., Li, M., Nguyen, J.T., Kiso, Y., Gustchina, A., Wlodawer, A.(2010) J Mol Biology 401: 626-641
- PubMed: 20600105 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1016/j.jmb.2010.06.052
- Primary Citation Related Structures:
3LIN, 3LIQ, 3LIT, 3LIV, 3LIX, 3LIY - PubMed Abstract:
Human T-cell leukemia virus type 1 (HTLV-1) is a retrovirus associated with several serious diseases, such as adult T-cell leukemia and tropical spastic paraparesis/myelopathy. For a number of years, the protease (PR) encoded by HTLV-1 has been a target for designing antiviral drugs, but that effort was hampered by limited available structural information. We report a high-resolution crystal structure of HTLV-1 PR complexed with a statine-containing inhibitor, a significant improvement over the previously available moderate-resolution structure. We also report crystal structures of the complexes of HTLV-1 PR with five different inhibitors that are more compact and more potent. A detailed study of structure-activity relationships was performed to interpret in detail the influence of the polar and hydrophobic interactions between the inhibitors and the protease.
- Protein Structure Section, Macromolecular Crystallography Laboratory, National Cancer Institute at Frederick, Frederick, MD 21702, USA.
Organizational Affiliation: