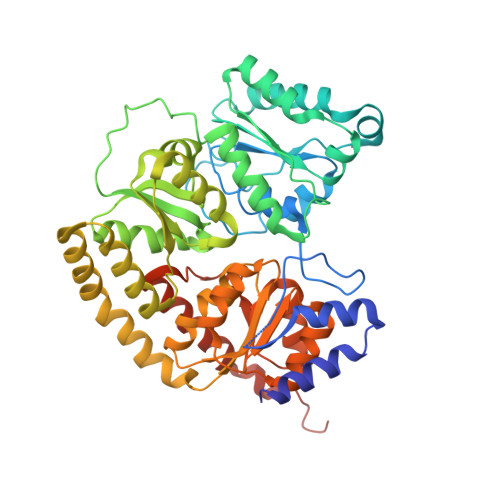
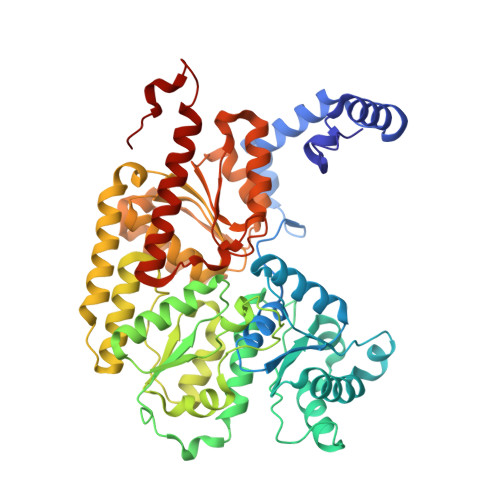
Insights into substrate binding at FeMo-cofactor in nitrogenase from the structure of an alpha-70(Ile) MoFe protein variant
Sarma, R., Barney, B.M., Keable, S., Dean, D.R., Seefeldt, L.C., Peters, J.W.(2010) J Inorg Biochem 104: 385-389
- PubMed: 20022118 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1016/j.jinorgbio.2009.11.009
- Primary Citation Related Structures:
3K1A - PubMed Abstract:
The X-ray crystal structure is presented for a nitrogenase MoFe protein where the alpha subunit residue at position 70 (alpha-70(Val)) has been substituted by the amino acid isoleucine (alpha-70(Ile)). Substitution of alpha-70(Val) by alpha-70(Ile) results in a MoFe protein that is hampered in its ability to reduce a range of substrates including acetylene and N(2), yet retains normal proton reduction activity. The 2.3A structure of the alpha-70(Ile) MoFe protein is compared to the alpha-70(Val) wild-type MoFe protein, revealing that the delta methyl group of alpha-70(Val) is positioned over Fe6 within the active site FeMo-cofactor. This work provides strong crystallographic support for the previously proposed model that substrates bind and are reduced at a single 4Fe-4S face of the FeMo-cofactor and that when alpha-70(Val) is substituted by alpha-70(Ile) access of substrates to Fe6 of this face is effectively blocked. Furthermore the detailed examination of the structure provides the basis for understanding the ability to trap and characterize hydrides in the variant, contributing significantly to our understanding of substrate access and substrate reduction at the FeMo-cofactor active site of nitrogenase.
- Department of Chemistry and Biochemistry and the Astrobiology Biogeocatalysis Research Center, Montana State University, Bozeman, MT 59717, USA.
Organizational Affiliation: