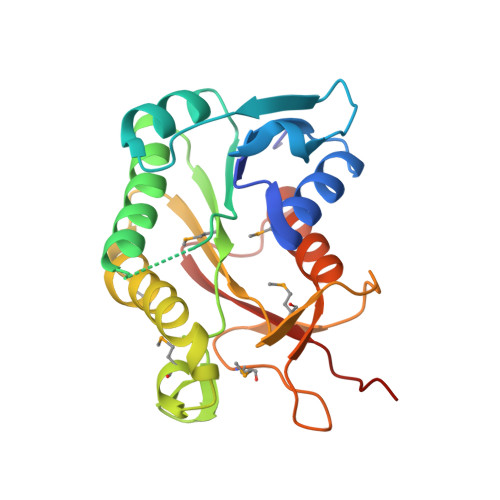
Crystal structure of the putative epimerase Q89Z24_BACTN from Bacteroides thetaiotaomicron.
Vorobiev, S.M., Su, M., Seetharaman, J., Wang, D., Ciccosanti, C., Foote, L.E., Janjua, H., Xiao, R., Acton, T., Montelione, G.T., Tong, L., Hunt, J.F.To be published.