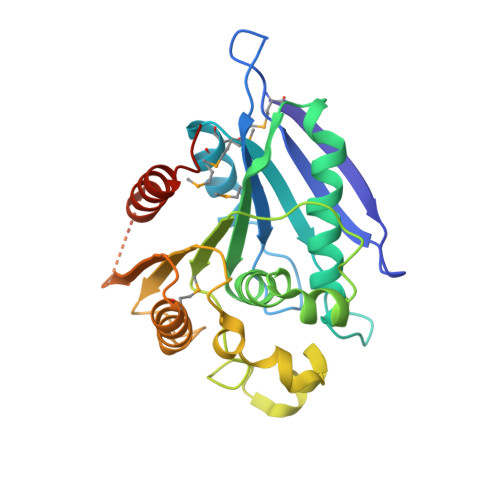
Crystal structure of lipase/esterase (lp_2923) from Lactobacillus plantarum. Northeast Structural Genomics Consortium target LpR108
Forouhar, F., Su, M., Seetharaman, J., Mao, L., Janjua, H., Xiao, R., Ciccosanti, C., Maglaqui, M., Foote, E.L., Zhao, L., Everett, J.K., Acton, T.B., Montelione, G.T., Tong, L., Hunt, J.F.To be published.