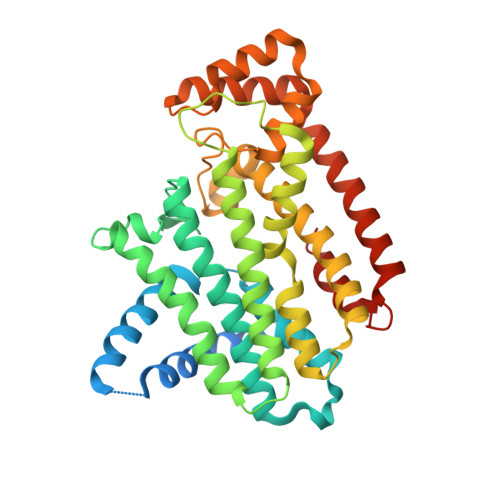
Human farnesyl diphosphate synthase (T201A mutant) complexed with Mg and biphosphonate inhibitor.
Pilka, E.S., Dunford, J.E., Guo, K., Pike, A.C.W., von Delft, F., Barnett, B.L., Ebetino, F.H., Arrowsmith, C.H., Bountra, C., Edwards, A.M., Russell, R.G.G., Oppermann, U.To be published.