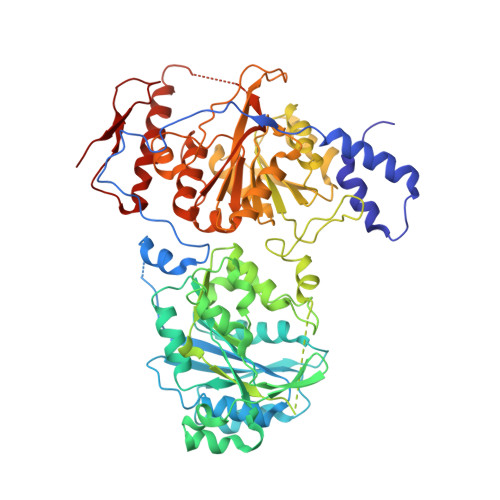
Crystal structure of chondroitin polymerase from Escherichia coli K4.
Osawa, T., Sugiura, N., Shimada, H., Hirooka, R., Tsuji, A., Shirakawa, T., Fukuyama, K., Kimura, M., Kimata, K., Kakuta, Y.(2009) Biochem Biophys Res Commun 378: 10-14
- PubMed: 18771653 Search on PubMed
- DOI: https://doi.org/10.1016/j.bbrc.2008.08.121
- Primary Citation Related Structures:
2Z86, 2Z87 - PubMed Abstract:
Elongation of glycosaminoglycan chains, such as heparan and chondroitin, is catalyzed by bi-functional glycosyltransferases, for which both 3-dimensional structures and reaction mechanisms remain unknown. The bacterial chondroitin polymerase K4CP catalyzes elongation of the chondroitin chain by alternatively transferring the GlcUA and GalNAc moiety from UDP-GlcUA and UDP-GalNAc to the non-reducing ends of the chondroitin chain. Here, we have determined the crystal structure of K4CP in the presence of UDP and UDP-GalNAc as well as with UDP and UDP-GlcUA. The structures consisted of two GT-A fold domains in which the two active sites were 60A apart. UDP-GalNAc and UDP-GlcUA were found at the active sites of the N-terminal and C-terminal domains, respectively. The present K4CP structures have provided the structural basis for further investigating the molecular mechanism of biosynthesis of chondroitin chain.
- Laboratory of Structural Biology, Graduate School of Systems Life Sciences, Kyushu University, Fukuoka 812-8581, Japan.
Organizational Affiliation: