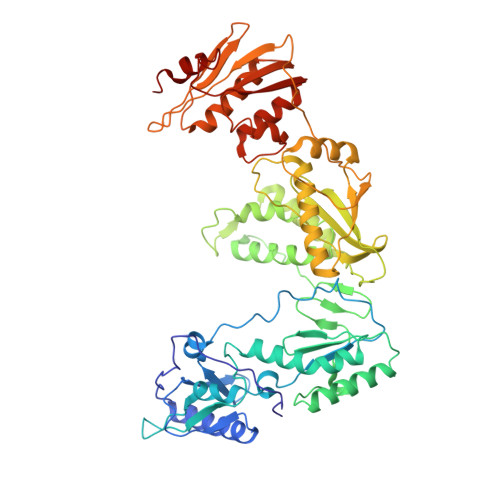
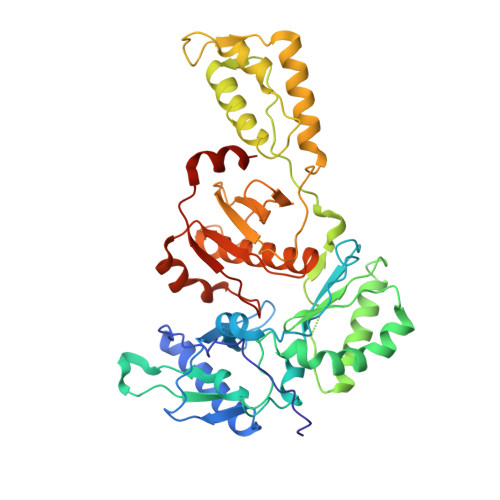
Lersivirine, a nonnucleoside reverse transcriptase inhibitor with activity against drug-resistant human immunodeficiency virus type 1.
Corbau, R., Mori, J., Phillips, C., Fishburn, L., Martin, A., Mowbray, C., Panton, W., Smith-Burchnell, C., Thornberry, A., Ringrose, H., Knochel, T., Irving, S., Westby, M., Wood, A., Perros, M.(2010) Antimicrob Agents Chemother 54: 4451-4463
- PubMed: 20660667 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1128/AAC.01455-09
- Primary Citation Related Structures:
2WOM, 2WON - PubMed Abstract:
The nonnucleoside reverse transcriptase inhibitors (NNRTIs) are key components of highly active antiretroviral therapy (HAART) for the treatment of human immunodeficiency virus type 1 (HIV-1). A major problem with the first approved NNRTIs was the emergence of mutations in the HIV-1 reverse transcriptase (RT), in particular K103N and Y181C, which led to resistance to the entire class. We adopted an iterative strategy to synthesize and test small molecule inhibitors from a chemical series of pyrazoles against wild-type (wt) RT and the most prevalent NNRTI-resistant mutants. The emerging candidate, lersivirine (UK-453,061), binds the RT enzyme in a novel way (resulting in a unique resistance profile), inhibits over 60% of viruses bearing key RT mutations, with 50% effective concentrations (EC(50)s) within 10-fold of those for wt viruses, and has excellent selectivity against a range of human targets. Altogether lersivirine is a highly potent and selective NNRTI, with excellent efficacy against NNRTI-resistant viruses.
- Department of Discovery Biology, Pfizer Global Research and Development, Sandwich, Kent CT13 9NJ, United Kingdom. romu.corbau@pfizer.com
Organizational Affiliation: