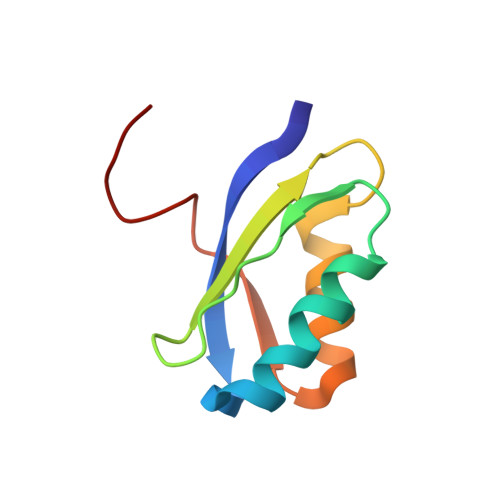
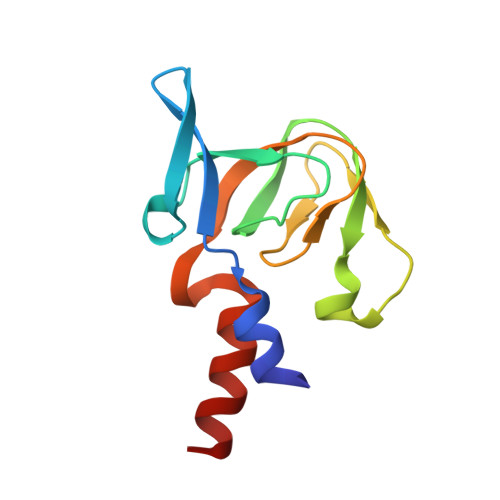
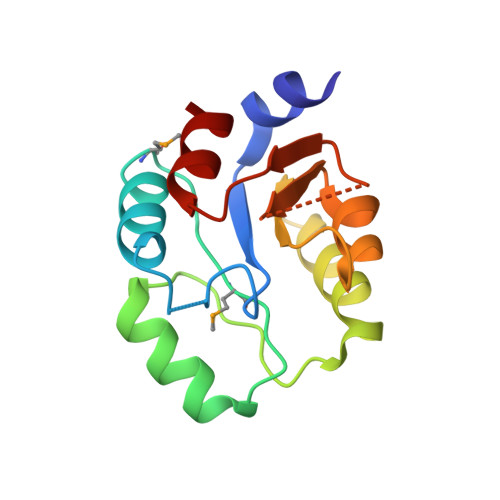
Structure of a Copper Pump Suggests a Regulatory Role for its Metal-Binding Domain.
Wu, C.-C., Rice, W.J., Stokes, D.L.(2008) Structure 16: 976
- PubMed: 18547529 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1016/j.str.2008.02.025
- Primary Citation Related Structures:
2VOY - PubMed Abstract:
P-type ATPases play an important role in Cu homeostasis, which provides sufficient Cu for metalloenzyme biosynthesis but prevents oxidative damage of free Cu to the cell. The P(IB) group of P-type ATPases includes ATP-dependent pumps of Cu and other transition metal ions, and it is distinguished from other family members by the presence of N-terminal metal-binding domains (MBD). We have determined structures of two constructs of a Cu pump from Archaeoglobus fulgidus (CopA) by cryoelectron microscopy of tubular crystals, which reveal the overall architecture and domain organization of the molecule. By comparing these structures, we localized its N-terminal MBD within the cytoplasmic domains that use ATP hydrolysis to drive the transport cycle. We have built a pseudoatomic model by fitting existing crystallographic structures into the cryoelectron microscopy maps for CopA, which suggest a Cu-dependent regulatory role for the MBD.
- Skirball Institute of Biomolecular Medicine, School of Medicine, New York University, 540 First Avenue, New York, NY 10016, USA.
Organizational Affiliation: