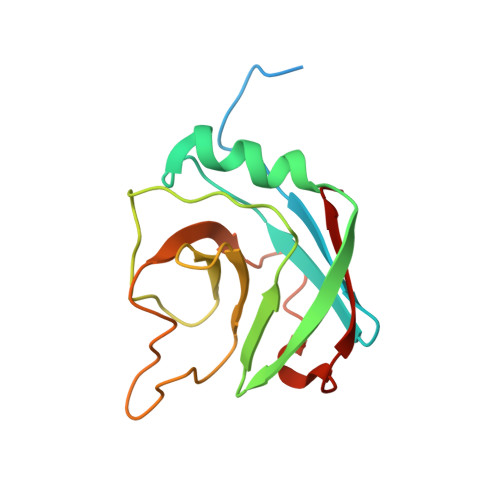
Hypothetical protein AF2241 from Archaeoglobus fulgidus adopts a cyclophilin-like fold.
Ai, X., Li, L., Semesi, A., Yee, A., Arrowsmith, C.H., Li, S.S., Choy, W.Y.(2007) J Biomol NMR 38: 353-358
- PubMed: 17610131 Search on PubMed
- DOI: https://doi.org/10.1007/s10858-007-9172-8
- Primary Citation Related Structures:
2NNZ - PubMed Abstract:
AF2241 is a hypothetical protein from Archaeoglobus fulgidus and it belongs to the PFam domain of unknown function 369 (DUF369). NMR structural determination reveals that AF2241 adopts a cyclophilin-like fold, with a beta-barrel core composed of eight beta-strands, one alpha-helix, and one 3(10) helix located at each end of the barrel. The protein displays a high structural similarity to TM1367, another member of DUF369 whose structure has been determined recently by X-ray crystallography. Structural similarity search shows that AF2241 also has a high similarity to human cyclophilin A, however, sequence alignment and electrostatic potential analysis reveal that the residues in the PPIase catalytic site of human cyclophilin A are not conserved in AF2241 or TM1367. Instead, a putative active site of AF2241 maps to a negatively charged pocket composed of 9 conserved residues. Our results suggest that although AF2241 adopts the same fold as the human cyclophilin A, it may have distinct biological function.
- Department of Biochemistry, The University of Western Ontario, London, Ontario, Canada.
Organizational Affiliation: