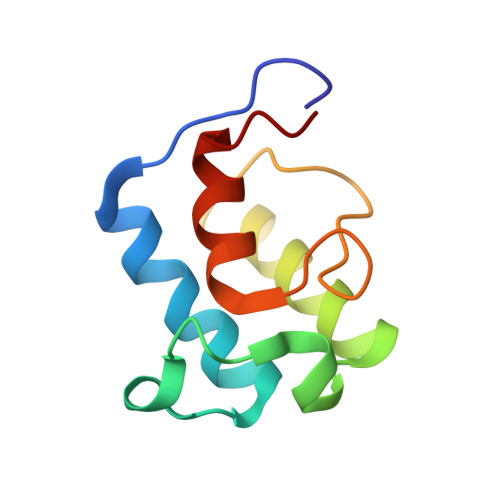
Solution structure of an acyl carrier protein domain from a fungal type I polyketide synthase.
Wattana-amorn, P., Williams, C., Ploskon, E., Cox, R.J., Simpson, T.J., Crosby, J., Crump, M.P.(2010) Biochemistry 49: 2186-2193
- PubMed: 20136099 Search on PubMed
- DOI: https://doi.org/10.1021/bi902176v
- Primary Citation Related Structures:
2KR5 - PubMed Abstract:
Acyl (peptidyl) carrier protein (ACP or PCP) is a crucial component involved in the transfer of thiol ester-bound intermediates during the biosynthesis of primary and secondary metabolites such as fatty acids, polyketides, and nonribosomal peptides. Although many carrier protein three-dimensional structures have been determined, to date there is no model available for a fungal type I polyketide synthase ACP. Here we report the solution structure of the norsolorinic acid synthase (NSAS) holo ACP domain that has been excised from the full-length multifunctional enzyme. NSAS ACP shows similarities in three-dimensional structure with other type I and type II ACPs, consisting of a four-helix bundle with helices I, II, and IV arranged in parallel. The N-terminus of helix III, however, is unusually hydrophobic, and Phe1768 and Leu1770 pack well with the core of the protein. The result is that unlike other carrier proteins, helix III lies almost perpendicular to the three major helices. Helix III is well-defined by numerous NMR-derived distance restraints and may be less flexible than counterparts in type II FAS and PKS ACPs. When the holo ACP is derivatized with a hexanoyl group, only minor changes are observed between the HSQC spectra of the two ACP species and no NOEs are observed for this hydrophobic acyl group. Along with the mammalian type I FAS, this further strengthens the view that type I ACPs do not show any significant affinity for hydrophobic (nonpolar) chain assembly intermediates attached via the 4'-phosphopantetheine prosthetic group.
- School of Chemistry, University of Bristol, Cantock's Close, Bristol BS8 1TS, UK.
Organizational Affiliation: