Crystal Structure of Human Prolidase: The Molecular Basis of Pd Disease
Mueller, U., Niesen, F.H., Roske, Y., Goetz, F., Behlke, J., Buessow, K., Heinemann, U.To be published.
Experimental Data Snapshot
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| XAA-PRO DIPEPTIDASE | 494 | Homo sapiens | Mutation(s): 0 EC: 3.4.13.9 | 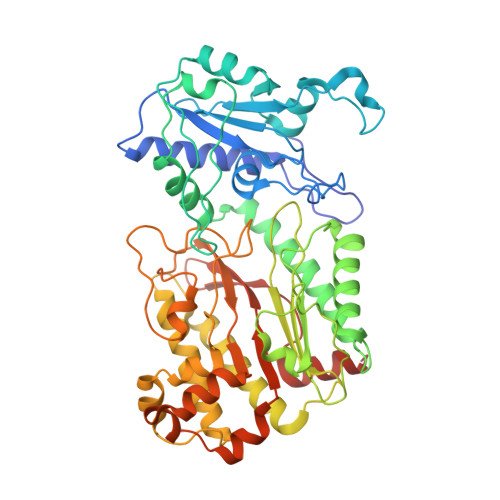 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P12955 GTEx: ENSG00000124299 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P12955 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| NA Download:Ideal Coordinates CCD File | C [auth A], D [auth A], E [auth A], F [auth B], G [auth B] | SODIUM ION Na FKNQFGJONOIPTF-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 103.579 | α = 90 |
| b = 108.518 | β = 90 |
| c = 211.024 | γ = 90 |
| Software Name | Purpose |
|---|---|
| REFMAC | refinement |
| DENZO | data reduction |
| SCALEPACK | data scaling |
| SOLVE/RESOLVE | phasing |