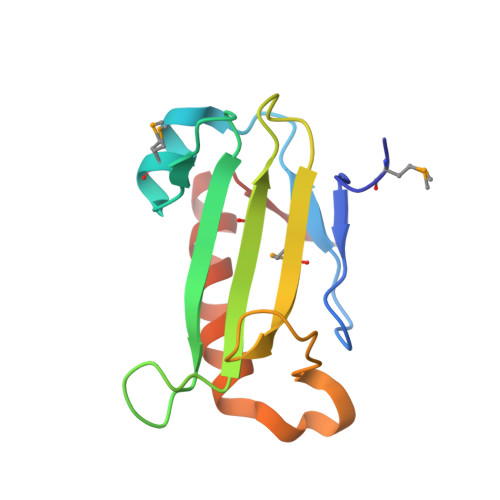
Structure of LP2179, the first representative of Pfam family PF08866, suggests a new fold with a role in amino-acid metabolism.
Bakolitsa, C., Kumar, A., Carlton, D., Miller, M.D., Krishna, S.S., Abdubek, P., Astakhova, T., Axelrod, H.L., Chiu, H.J., Clayton, T., Deller, M.C., Duan, L., Elsliger, M.A., Feuerhelm, J., Grzechnik, S.K., Grant, J.C., Han, G.W., Jaroszewski, L., Jin, K.K., Klock, H.E., Knuth, M.W., Kozbial, P., Marciano, D., McMullan, D., Morse, A.T., Nigoghossian, E., Okach, L., Oommachen, S., Paulsen, J., Reyes, R., Rife, C.L., Tien, H.J., Trout, C.V., van den Bedem, H., Weekes, D., Xu, Q., Hodgson, K.O., Wooley, J., Deacon, A.M., Godzik, A., Lesley, S.A., Wilson, I.A.(2010) Acta Crystallogr Sect F Struct Biol Cryst Commun 66: 1205-1210
- PubMed: 20944212 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1107/S1744309109023689
- Primary Citation Related Structures:
2IAY - PubMed Abstract:
The structure of LP2179, a member of the PF08866 (DUF1831) family, suggests a novel α+β fold comprising two β-sheets packed against a single helix. A remote structural similarity to two other uncharacterized protein families specific to the Bacillus genus (PF08868 and PF08968), as well as to prokaryotic S-adenosylmethionine decarboxylases, is consistent with a role in amino-acid metabolism. Genomic neighborhood analysis of LP2179 supports this functional assignment, which might also then be extended to PF08868 and PF08968.
- Stanford Synchrotron Radiation Lightsource, SLAC National Accelerator Laboratory, Menlo Park, CA, USA.
Organizational Affiliation: