Structure and Function of Cyclized Versions of the Proofreading Exonuclease Subunit E. coli DNA Polymerase III
Park, A.Y., Carr, P.D., Oliis, D.L., Dixon, N.E.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| DNA polymerase III epsilon subunit | 194 | Escherichia coli | Mutation(s): 0 Gene Names: dnaQ, mutD EC: 2.7.7.7 | 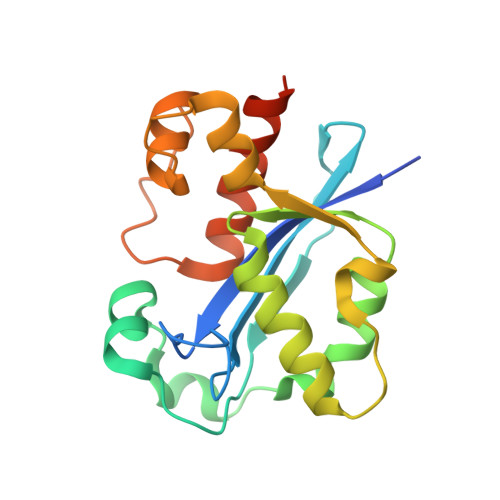 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P03007 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 4 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| U5P Download:Ideal Coordinates CCD File | D [auth A], E [auth A] | URIDINE-5'-MONOPHOSPHATE C9 H13 N2 O9 P DJJCXFVJDGTHFX-XVFCMESISA-N |  | ||
| PEG Download:Ideal Coordinates CCD File | H [auth A] | DI(HYDROXYETHYL)ETHER C4 H10 O3 MTHSVFCYNBDYFN-UHFFFAOYSA-N |  | ||
| EDO Download:Ideal Coordinates CCD File | F [auth A], G [auth A] | 1,2-ETHANEDIOL C2 H6 O2 LYCAIKOWRPUZTN-UHFFFAOYSA-N |  | ||
| MN Download:Ideal Coordinates CCD File | B [auth A], C [auth A] | MANGANESE (II) ION Mn WAEMQWOKJMHJLA-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 60.099 | α = 90 |
| b = 60.099 | β = 90 |
| c = 109.164 | γ = 90 |
| Software Name | Purpose |
|---|---|
| DENZO | data reduction |
| SCALEPACK | data scaling |
| REFMAC | refinement |
| PDB_EXTRACT | data extraction |