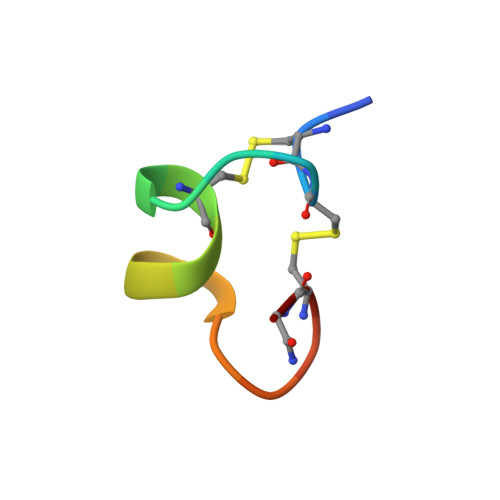
Solution conformation of a neuronal nicotinic acetylcholine receptor antagonist alpha-conotoxin OmIA that discriminates alpha3 vs. alpha6 nAChR subtypes
Chi, S.-W., Kim, D.-H., Olivera, B.M., McIntosh, J.M., Han, K.-H.(2006) Biochem Biophys Res Commun 345: 248-254
- PubMed: 16678128 Search on PubMed
- DOI: https://doi.org/10.1016/j.bbrc.2006.04.099
- Primary Citation Related Structures:
2GCZ - PubMed Abstract:
alpha-Conotoxin OmIA from Conus omaria is the only alpha-conotoxin that shows a approximately 20-fold higher affinity to the alpha3beta2 over the alpha6beta2 subtype of nicotinic acetylcholine receptor. We have determined a three-dimensional structure of alpha-conotoxin OmIA by nuclear magnetic resonance spectroscopy. alpha-Conotoxin OmIA has an "omega-shaped" overall topology with His(5)-Asn(12) forming an alpha-helix. Structural features of alpha-conotoxin OmIA responsible for its selectivity are suggested by comparing its surface characteristics with other functionally related alpha4/7 subfamily conotoxins. Reduced size of the hydrophilic area in alpha-conotoxin OmIA seems to be associated with the reduced affinity towards the alpha6beta2 nAChR subtype.
- Molecular Anti-Cancer Research Center, Division of Molecular Therapeutics, Korea Research Institute of Bioscience and Biotechnology, Yusong P.O. Box 115, Daejon, Republic of Korea.
Organizational Affiliation: