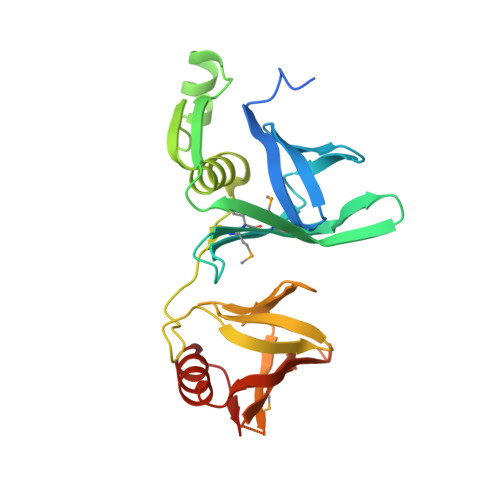
The Structure of the yFACT Pob3-M Domain, Its Interaction with the DNA Replication Factor RPA, and a Potential Role in Nucleosome Deposition.
VanDemark, A.P., Blanksma, M., Ferris, E., Heroux, A., Hill, C.P., Formosa, T.(2006) Mol Cell 22: 363-374
- PubMed: 16678108 Search on PubMed
- DOI: https://doi.org/10.1016/j.molcel.2006.03.025
- PubMed Abstract:
We report the crystal structure of the middle domain of the Pob3 subunit (Pob3-M) of S. cerevisiae FACT (yFACT, facilitates chromatin transcription), which unexpectedly adopts an unusual double pleckstrin homology (PH) architecture. A mutation within a conserved surface cluster in this domain causes a defect in DNA replication that is suppressed by mutation of replication protein A (RPA). The nucleosome reorganizer yFACT therefore interacts in a physiologically important way with the central single-strand DNA (ssDNA) binding factor RPA to promote a step in DNA replication. Purified yFACT and RPA display a weak direct physical interaction, although the genetic suppression is not explained by simple changes in affinity between the purified proteins. Further genetic analysis suggests that coordinated function by yFACT and RPA is important during nucleosome deposition. These results support the model that the FACT family has an essential role in constructing nucleosomes during DNA replication, and suggest that RPA contributes to this process.
- Department of Biochemistry, University of Utah School of Medicine, Salt Lake City, 84132, USA.
Organizational Affiliation: