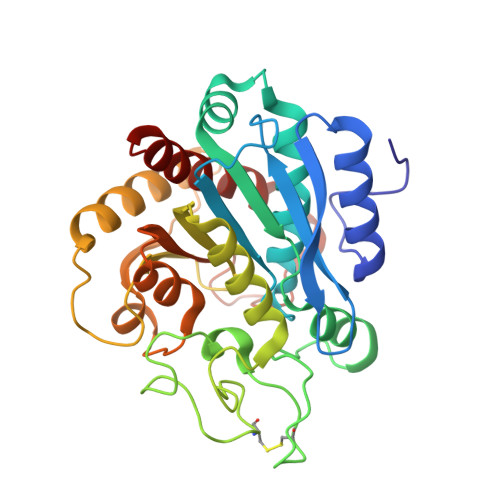
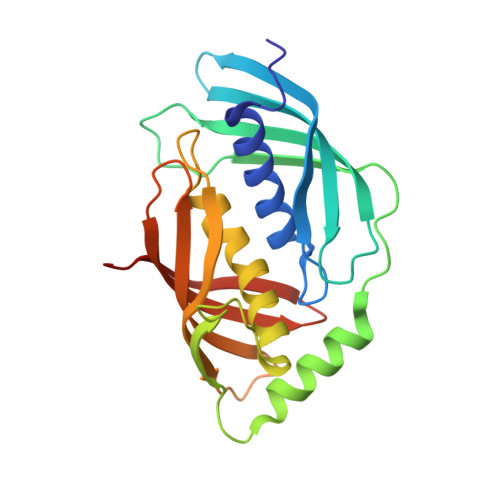
Structure of Human Carboxypeptidase A4 with its Endogenous Protein Inhibitor, Latexin.
Pallares, I., Bonet, R., Garcia-Castellanos, R., Ventura, S., Aviles, F.X., Vendrell, J., Gomis-Rueth, F.X.(2005) Proc Natl Acad Sci U S A 102: 3978
- PubMed: 15738388 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1073/pnas.0500678102
- Primary Citation Related Structures:
2BO9 - PubMed Abstract:
The only endogenous protein inhibitor known for metallocarboxypeptidases (MCPs) is latexin, a 25-kDa protein discovered in the rat brain. Latexin, alias endogenous carboxypeptidase inhibitor, inhibits human CPA4 (hCPA4), whose expression is induced in prostate cancer cells after treatment with histone deacetylase inhibitors. hCPA4 is a member of the A/B subfamily of MCPs and displays the characteristic alpha/beta-hydrolase fold. Human latexin consists of two topologically equivalent subdomains, reminiscent of cystatins, consisting of an alpha-helix enveloped by a curved beta-sheet. These subdomains are packed against each other through the helices and linked by a connecting segment encompassing a third alpha-helix. The enzyme is bound at the interface of these subdomains. The complex occludes a large contact surface but makes rather few contacts, despite a nanomolar inhibition constant. This low specificity explains the flexibility of latexin in inhibiting all vertebrate A/B MCPs tested, even across species barriers. In contrast, modeling studies reveal why the N/E subfamily of MCPs and invertebrate A/B MCPs are not inhibited. Major differences in the loop segments shaping the border of the funnel-like access to the protease active site impede complex formation with latexin. Several sequences ascribable to diverse tissues and organs have been identified in vertebrate genomes as being highly similar to latexin. They are proposed to constitute the latexin family of potential inhibitors. Because they are ubiquitous, latexins could represent for vertebrate A/B MCPs the counterparts of tissue inhibitors of metalloproteases for matrix metalloproteinases.
- Institut de Biologia Molecular de Barcelona, Centro de Investigación y Desarrollo-Consejo Superior de Investigaciones Científicas, C/Jordi Girona, 18-26, E-08034 Barcelona, Spain.
Organizational Affiliation: