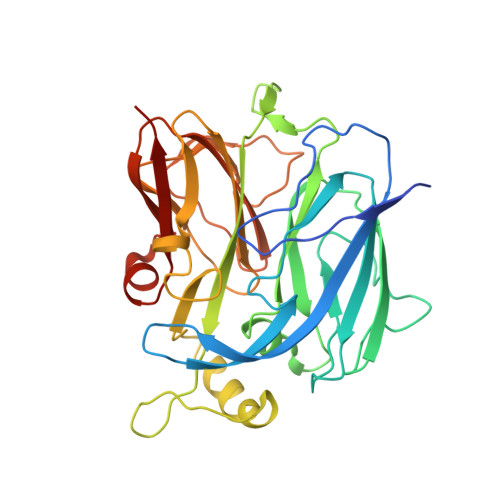
Crystal structure of C-terminal desundecapeptide nitrite reductase from Achromobacter cycloclastes
Li, H.T., Chang, T., Chang, W.C., Chen, C.J., Liu, M.Y., Gui, L.L., Zhang, J.P., An, X.M., Chang, W.R.(2005) Biochem Biophys Res Commun 338: 1935-1942
- PubMed: 16293231 Search on PubMed
- DOI: https://doi.org/10.1016/j.bbrc.2005.09.199
- Primary Citation Related Structures:
2AVF - PubMed Abstract:
Monoclinic crystal structure of C-terminal desundecapeptide nitrite reductase (NiRc-11) from Achromobacter cycloclastes was determined at 2.6A. NiRc-11 exists as a loose trimer in the crystal. Deletion of 11 residues eliminates all intersubunit hydrogen bonds mediated by the C-terminal tail. The rigid irregular coil 105-112, which constitutes part of the sidewall of the active site pocket, undergoes conformational changes and becomes highly flexible in NiRc-11. Correspondingly, the linker segments between the two copper sites 95-100 and 135-136 are partly relaxed in conformation, which leads to disrupted active site microenvironments responsible for the activity loss and spectral change of NiRc-11. Comparison with the native structure revealed a bulky residue Met331 fastened by hydrogen bonding, which may play a direct role in keeping the right copper site geometry by protruding its side chain against the irregular coil 105-112. Sequence alignment showed that the bulky residue is conserved at position 331, indicating an equal importance of C-terminal segment in other copper-containing nitrite reductases.
- National Laboratory of Biomacromolecules, Institute of Biophysics, Chinese Academy of Sciences, 15th Datun Road, Chaoyang District, Beijing 100101, China.
Organizational Affiliation: