Open-state structure of veratridine-activated human Nav1.7 reveals the molecular choreography of fast inactivation
Fan, X., Chen, J., Xue, L., Wang, H., Wu, T., Huang, X., Lu, F., Jin, X., Song, C., Huang, J., Yan, N.To be published.
Experimental Data Snapshot
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Sodium channel regulatory subunit beta-1 | A [auth B] | 173 | Homo sapiens | Mutation(s): 0 Gene Names: SCN1B | 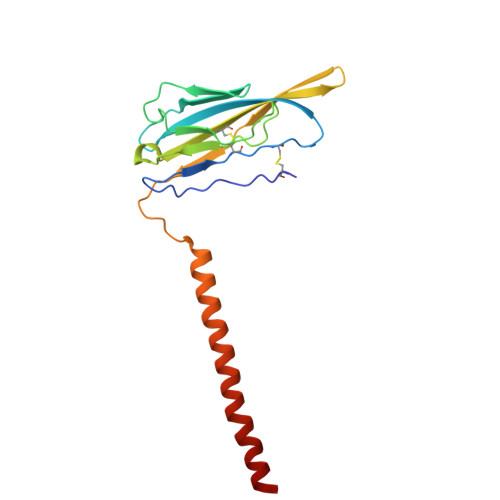 |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: Q07699 GTEx: ENSG00000105711 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q07699 | ||||
Glycosylation | |||||
| Glycosylation Sites: 4 | Go to GlyGen: Q07699-1 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Sodium channel regulatory subunit beta-2 | B [auth C] | 119 | Homo sapiens | Mutation(s): 0 Gene Names: SCN2B, UNQ326/PRO386 | 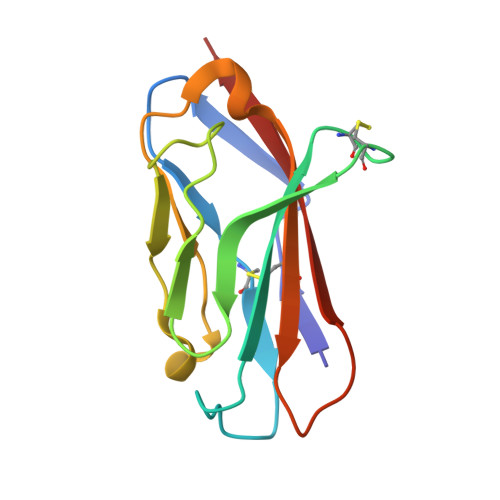 |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: O60939 GTEx: ENSG00000149575 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | O60939 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 3 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Sodium channel protein type 9 subunit alpha | C [auth A] | 1,988 | Homo sapiens | Mutation(s): 0 Gene Names: SCN9A, NENA | 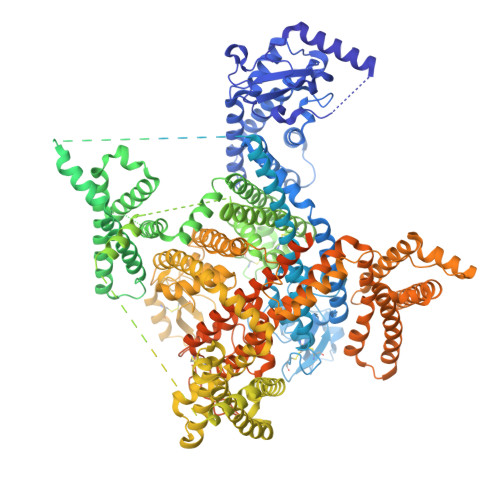 |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: Q15858 GTEx: ENSG00000169432 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q15858 | ||||
Glycosylation | |||||
| Glycosylation Sites: 4 | Go to GlyGen: Q15858-1 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 6 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| PCW Download:Ideal Coordinates CCD File | S [auth A], V [auth A] | 1,2-DIOLEOYL-SN-GLYCERO-3-PHOSPHOCHOLINE C44 H85 N O8 P SNKAWJBJQDLSFF-NVKMUCNASA-O |  | ||
| A1E26 (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | M [auth A] | Veratridine C36 H51 N O11 FVECELJHCSPHKY-YFUMOZOISA-N |  | ||
| P3X Download:Ideal Coordinates CCD File | AA [auth A] | (5E,17R,20S)-23-amino-20-hydroxy-14,20-dioxo-15,19,21-trioxa-20lambda~5~-phosphatricos-5-en-17-yl hexadecanoate C35 H68 N O8 P IXIBEFBSXYIWMP-VCRBCDDQSA-N |  | ||
| LPE Download:Ideal Coordinates CCD File | I [auth B] Q [auth A] R [auth A] T [auth A] U [auth A] | 1-O-OCTADECYL-SN-GLYCERO-3-PHOSPHOCHOLINE C26 H57 N O6 P XKBJVQHMEXMFDZ-AREMUKBSSA-O |  | ||
| Y01 Download:Ideal Coordinates CCD File | N [auth A], O [auth A], P [auth A], Z [auth A] | CHOLESTEROL HEMISUCCINATE C31 H50 O4 WLNARFZDISHUGS-MIXBDBMTSA-N |  | ||
| NAG Download:Ideal Coordinates CCD File | F [auth B] G [auth B] H [auth B] J [auth A] K [auth A] | 2-acetamido-2-deoxy-beta-D-glucopyranose C8 H15 N O6 OVRNDRQMDRJTHS-FMDGEEDCSA-N |  | ||
| Task | Software Package | Version |
|---|---|---|
| MODEL REFINEMENT | PHENIX | 1.20.1_4487 |
| RECONSTRUCTION | cryoSPARC | |
| RECONSTRUCTION | cryoSPARC | |
| RECONSTRUCTION | cryoSPARC |
| Funding Organization | Location | Grant Number |
|---|---|---|
| Human Frontier Science Program (HFSP) | France | LT000754/2020-L |